
Journal of Inorganic Materials ›› 2022, Vol. 37 ›› Issue (9): 991-1000.DOI: 10.15541/jim20210638
• RESEARCH ARTICLE • Previous Articles Next Articles
WANG Hongning1( ), HUANG Li1, QING Jiang3, MA Tengzhou3(
), HUANG Li1, QING Jiang3, MA Tengzhou3( ), HUANG Weiqiu2, CHEN Ruoyu1(
), HUANG Weiqiu2, CHEN Ruoyu1( )
)
Received:2021-10-18
Revised:2022-02-18
Published:2022-09-20
Online:2022-03-15
Contact:
CHEN Ruoyu, professor. E-mail: chry@cczu.edu.cn;About author:WANG Hongning (1980-), female, PhD candidate. E-mail: 444873772@qq.com
Supported by:CLC Number:
WANG Hongning, HUANG Li, QING Jiang, MA Tengzhou, HUANG Weiqiu, CHEN Ruoyu. Mesoporous Organic-inorganic Hybrid Siliceous Hollow Spheres: Synthesis and VOCs Adsorption[J]. Journal of Inorganic Materials, 2022, 37(9): 991-1000.
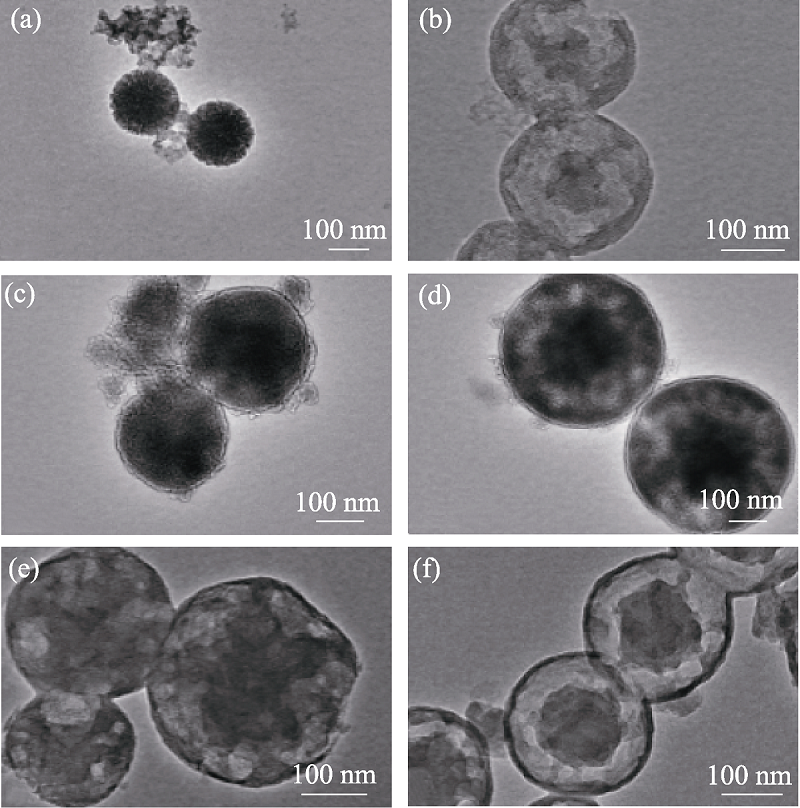
Fig. 1 TEM images of MOSs with different initial BTSE/ (BTSE+TEOS) molar ratios (a) MOS-0; (b) MOS-5%; (c) MOS-7.5%; (d) MOS-10%; (e) MOS-12.5%; (f) MOS-15%
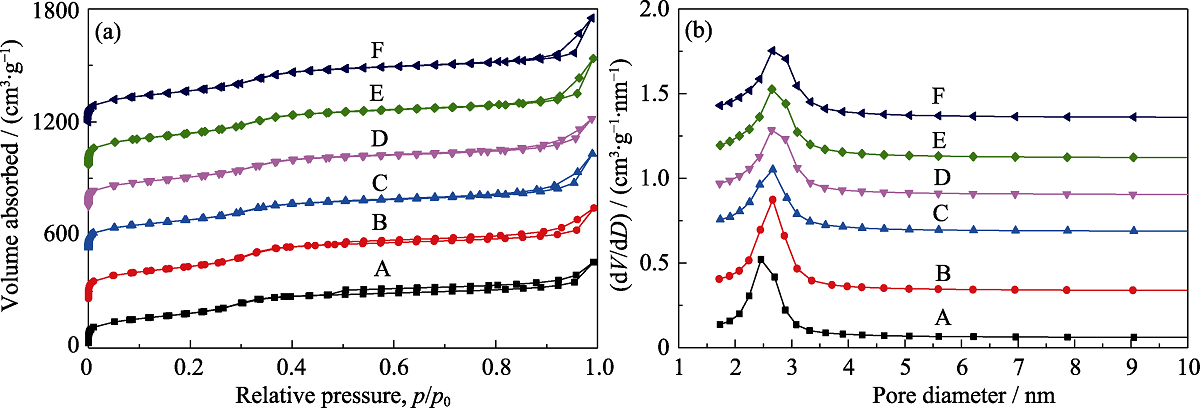
Fig. 2 N2 sorption isotherms (a) and pore size distributions (b) of MOSs with different initial BTSE/(BTSE+TEOS) molar ratios (A) MOS-0; (B) MOS-5%; (C) MOS-7.5%; (D) MOS-10%; (E)MOS-12.5%; (F) MOS-15%. In (a), the Y-axis values of (B-F) are 300, 600, 800, 1000, and 1300 m2·g-1, respectively. In (b), the Y-axis values of (B-F) are 0.1, 0.4, 0.8, 1.0, 1.2, and 1.4 cm3·g-1, respectively
| Sample | SBET/ (m2·g-1) | Sm/ (m2·g-1) | Vt/ (cm3·g-1) | Vm/ (cm3·g-1) | Pore size/ nm |
|---|---|---|---|---|---|
| MOS-0 | 591 | 0 | 0.721 | 0 | 2.5 |
| MOS-5% | 612 | 0 | 0.722 | 0 | 2.7 |
| MOS-7.5% | 612 | 0 | 0.776 | 0 | 2.8 |
| MOS-10% | 696 | 0 | 0.887 | 0 | 2.6 |
| MOS-12.5% | 655 | 0 | 0.873 | 0 | 2.7 |
| MOS-15% | 648 | 0 | 0.857 | 0 | 2.7 |
Table 1 Structural parameters of MOSs with different initial BTSE/(BTSE+TEOS) molar ratios
| Sample | SBET/ (m2·g-1) | Sm/ (m2·g-1) | Vt/ (cm3·g-1) | Vm/ (cm3·g-1) | Pore size/ nm |
|---|---|---|---|---|---|
| MOS-0 | 591 | 0 | 0.721 | 0 | 2.5 |
| MOS-5% | 612 | 0 | 0.722 | 0 | 2.7 |
| MOS-7.5% | 612 | 0 | 0.776 | 0 | 2.8 |
| MOS-10% | 696 | 0 | 0.887 | 0 | 2.6 |
| MOS-12.5% | 655 | 0 | 0.873 | 0 | 2.7 |
| MOS-15% | 648 | 0 | 0.857 | 0 | 2.7 |
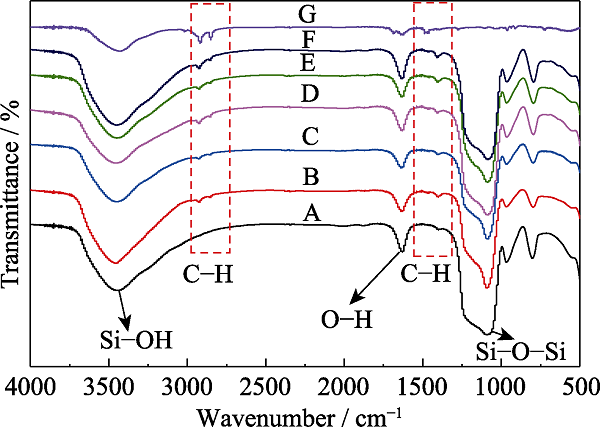
Fig. 3 FT-IR spectra of MOSs with different initial BTSE/ (BTSE+TEOS) molar ratios (A) MOS-0; (B) MOS-5%; (C) MOS-7.5%; (D) MOS-10%; (E) MOS- 12.5%; (F) MOS-15%; (G) CTAB
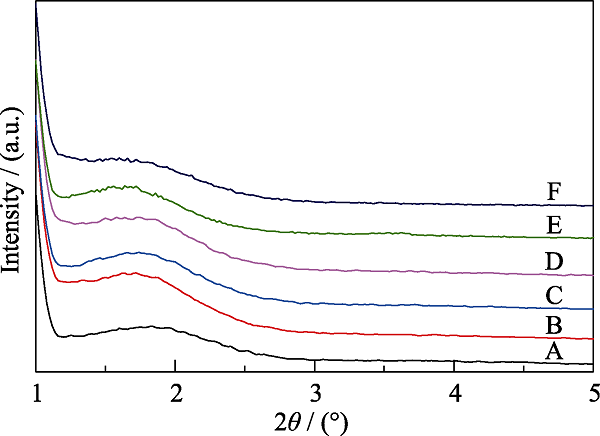
Fig. 4 XRD patterns of MOSs with different initial BTSE/ (BTSE+TEOS) molar ratios (A) MOS-0; (B) MOS-5%; (C) MOS-7.5%; (D) MOS-10%; (E) MOS- 12.5%; (F) MOS-15%
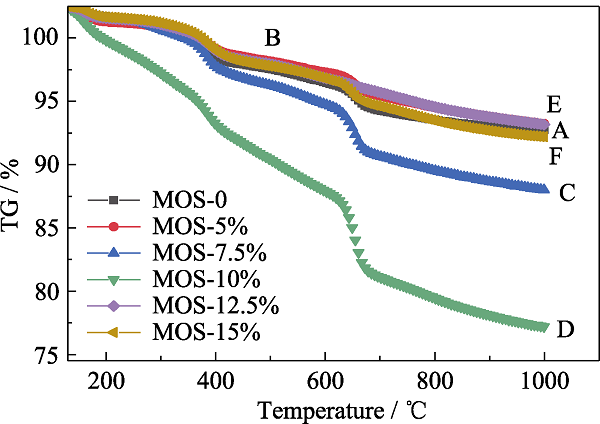
Fig. 5 TGA curves of MOSs with different initial BTSE/ (BTSE+TEOS) molar ratios (A) MOS-0; (B) MOS-5%; (C) MOS-7.5%; (D) MOS-10%; (E) MOS- 12.5%; (F) MOS-15%;

Fig. 6 Histograms of static VOCs (n-hexane, toluene and 92# gasoline) adsorption capacities (a, c, e) and desorption efficiencies (b, d, f) of different samples (A) MOS-0; (B) MOS-5%; (C) MOS-7.5%; (D) MOS-10%; (E) MOS-12.5%; (F) MOS-15%; (G) SG; (H) AC
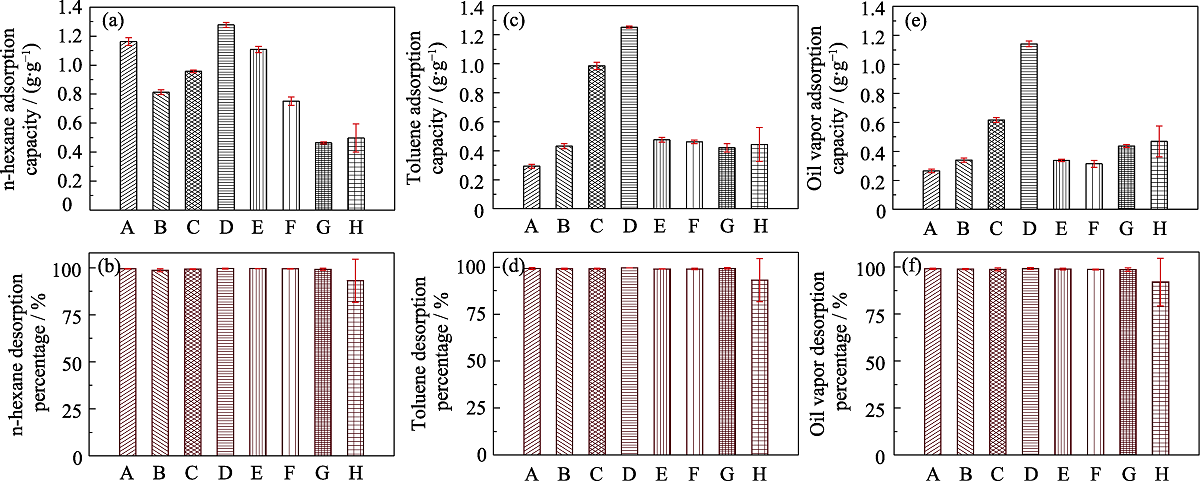
Fig. 6 Histograms of static VOCs (n-hexane, toluene and 92# gasoline) adsorption capacities (a, c, e) and desorption efficiencies (b, d, f) of different samples (A) MOS-0; (B) MOS-5%; (C) MOS-7.5%; (D) MOS-10%; (E) MOS-12.5%; (F) MOS-15%; (G) SG; (H) AC
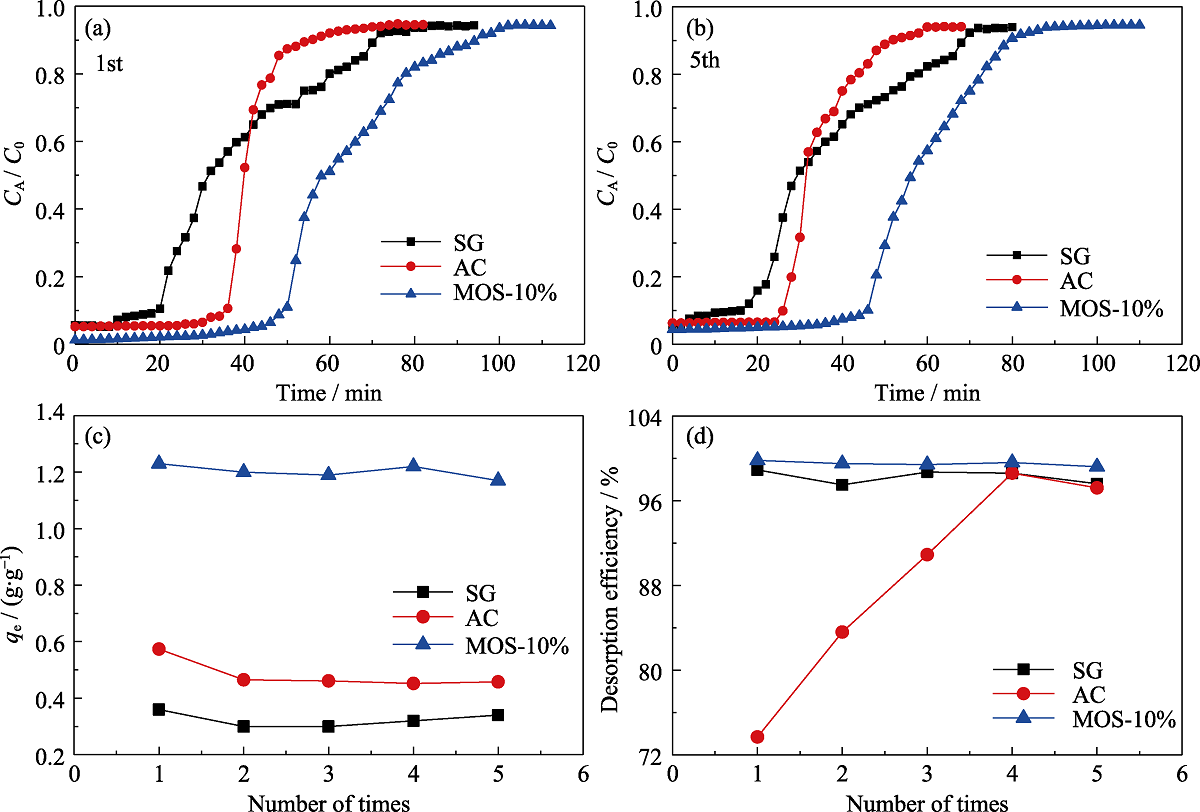
Fig. 8 Breakthrough curves for n-hexane of SG (■), AC (●) and MOS-10% (▲) under dry condition for the (a) first time and (b) fifth time and comparison of the qe (c) and desorption efficiency (d) of 5 times
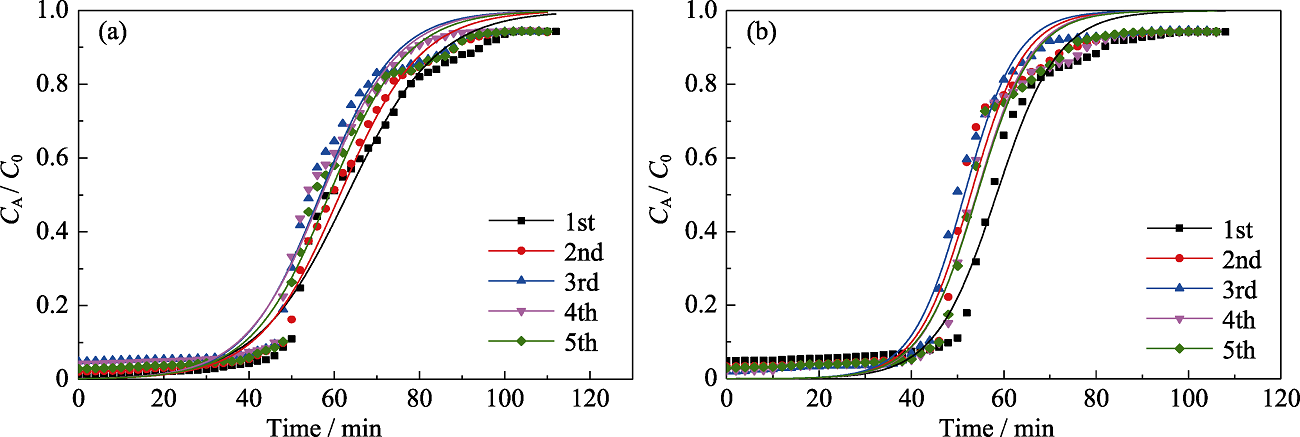
Fig. 10 Yoon and Nelson model fitting for 5 times adsorption of n-hexane (a) and toluene (b) on MOS-10% under dry condition Colorful figures are available on website
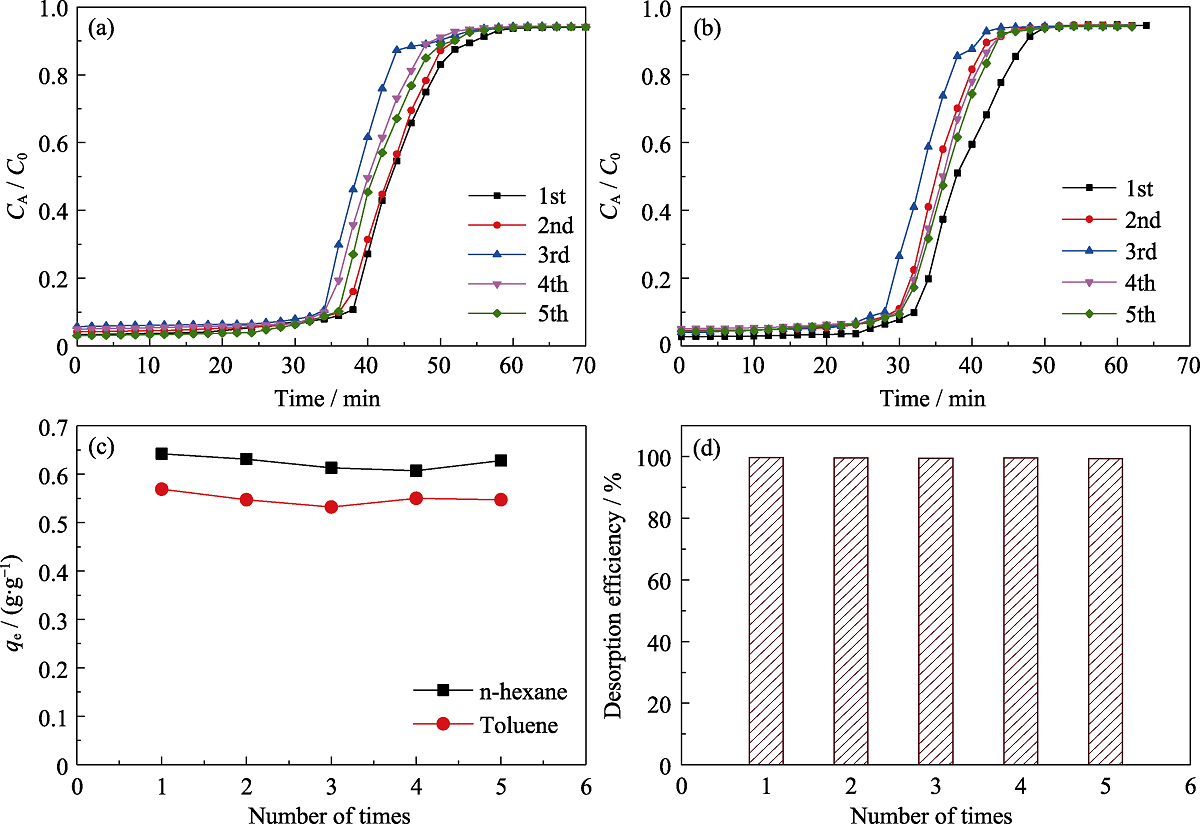
Fig. 12 Simultaneous breakthrough adsorption n-hexane (a) and toluene (b) on MOS-10% under dry condition, comparison of the equilibrium adsorption capacities (c) and desorption efficiency (d) for 5 times, respectively
| Sample | SBET/(m2 ·g-1) | Sm/(m2 ·g-1) | Vt/(cm3 ·g-1) | Vm/(cm3 ·g-1) | Pore size/nm |
|---|---|---|---|---|---|
| AC | 1451 | 973 | 1.03 | 0.48 | 5.6 |
| SG | 430 | 15 | 0.710 | 0.010 | 6.9 |
Table S1 Structural parameters of AC and SG
| Sample | SBET/(m2 ·g-1) | Sm/(m2 ·g-1) | Vt/(cm3 ·g-1) | Vm/(cm3 ·g-1) | Pore size/nm |
|---|---|---|---|---|---|
| AC | 1451 | 973 | 1.03 | 0.48 | 5.6 |
| SG | 430 | 15 | 0.710 | 0.010 | 6.9 |
| Sample | First event | Second event | Residual mass /% | ||
|---|---|---|---|---|---|
| (Tons-Tf )/℃ | Δm/% | (Tons-Tf )/℃ | Δm/% | ||
| MOS-0 | 30-200 | 1.7 | 200-900 | 8.1 | 90.2 |
| MOS-5% | 30-200 | 1.7 | 200-900 | 7.6 | 90.7 |
| MOS-7.5% | 30-200 | 1.9 | 200-900 | 12.6 | 85.5 |
| MOS-10% | 30-200 | 5.3 | 200-900 | 20.1 | 74.6 |
| MOS-12.5% | 30-200 | 1.5 | 200-900 | 7.9 | 90.6 |
| MOS-15% | 30-200 | 1.4 | 200-900 | 9.0 | 89.6 |
Table S2 TGA results of different MOSs under nitrogen atmosphere in the range of 30-900 ℃ (10 ℃/min)
| Sample | First event | Second event | Residual mass /% | ||
|---|---|---|---|---|---|
| (Tons-Tf )/℃ | Δm/% | (Tons-Tf )/℃ | Δm/% | ||
| MOS-0 | 30-200 | 1.7 | 200-900 | 8.1 | 90.2 |
| MOS-5% | 30-200 | 1.7 | 200-900 | 7.6 | 90.7 |
| MOS-7.5% | 30-200 | 1.9 | 200-900 | 12.6 | 85.5 |
| MOS-10% | 30-200 | 5.3 | 200-900 | 20.1 | 74.6 |
| MOS-12.5% | 30-200 | 1.5 | 200-900 | 7.9 | 90.6 |
| MOS-15% | 30-200 | 1.4 | 200-900 | 9.0 | 89.6 |
| Sample | Adsorption | Desorption | -OH/(×1020, g-1) | ||
|---|---|---|---|---|---|
| Average/(g·g-1) | STDEa/% | Average/% | STDEa/% | ||
| MOS-0% | 0.903 | 0.0168 | 99.4 | 0.205 | 3.11 |
| MOS-5% | 0.667 | 0.0177 | 99.3 | 0.329 | 2.35 |
| MOS-7.5% | 0.656 | 0.0165 | 99.4 | 0.283 | 2.31 |
| MOS-10% | 0.630 | 0.0137 | 99.4 | 0.134 | 2.23 |
| MOS-12.5% | 0.697 | 0.0156 | 99.4 | 0.230 | 2.44 |
| MOS-15% | 0.697 | 0.0136 | 99.4 | 0.365 | 2.45 |
| SG | 0.433 | 0.0984 | 97.7 | 0.867 | 1.72 |
| AC | 0.482 | 0.0305 | 93.5 | 11.1 | 1.77 |
Table S3 Water vapor adsorption capacities, desorption efficiencies and the densities of surface hydroxyl groups of different samples
| Sample | Adsorption | Desorption | -OH/(×1020, g-1) | ||
|---|---|---|---|---|---|
| Average/(g·g-1) | STDEa/% | Average/% | STDEa/% | ||
| MOS-0% | 0.903 | 0.0168 | 99.4 | 0.205 | 3.11 |
| MOS-5% | 0.667 | 0.0177 | 99.3 | 0.329 | 2.35 |
| MOS-7.5% | 0.656 | 0.0165 | 99.4 | 0.283 | 2.31 |
| MOS-10% | 0.630 | 0.0137 | 99.4 | 0.134 | 2.23 |
| MOS-12.5% | 0.697 | 0.0156 | 99.4 | 0.230 | 2.44 |
| MOS-15% | 0.697 | 0.0136 | 99.4 | 0.365 | 2.45 |
| SG | 0.433 | 0.0984 | 97.7 | 0.867 | 1.72 |
| AC | 0.482 | 0.0305 | 93.5 | 11.1 | 1.77 |
| Sample | tb/min | te/min | qe/(g·g-1) | Desorption efficiency/% |
|---|---|---|---|---|
| MOS-10%-1st | 50 | 104 | 1.23 | 99.8 |
| MOS-10%-2nd | 48 | 102 | 1.20 | 99.5 |
| MOS-10%-3rd | 46 | 100 | 1.19 | 99.4 |
| MOS-10%-4th | 48 | 102 | 1.22 | 99.6 |
| MOS-10%-5th | 48 | 102 | 1.21 | 99.2 |
| SG-1st | 16 | 72 | 0.361 | 98.9 |
| SG-2nd | 14 | 70 | 0.321 | 97.5 |
| SG-3rd | 14 | 70 | 0.334 | 98.7 |
| SG-4th | 12 | 72 | 0.321 | 98.6 |
| SG-5th | 10 | 72 | 0.343 | 97.6 |
| AC-1st | 38 | 50 | 0.574 | 73.7 |
| AC-2nd | 28 | 44 | 0.465 | 83.6 |
| AC-3rd | 28 | 42 | 0.461 | 90.9 |
| AC-4th | 26 | 44 | 0.452 | 98.6 |
| AC-5th | 24 | 42 | 0.458 | 97.2 |
Table S4 Comparison of dynamic n-hexane adsorption parameters on different samples for 5 times under dry condition
| Sample | tb/min | te/min | qe/(g·g-1) | Desorption efficiency/% |
|---|---|---|---|---|
| MOS-10%-1st | 50 | 104 | 1.23 | 99.8 |
| MOS-10%-2nd | 48 | 102 | 1.20 | 99.5 |
| MOS-10%-3rd | 46 | 100 | 1.19 | 99.4 |
| MOS-10%-4th | 48 | 102 | 1.22 | 99.6 |
| MOS-10%-5th | 48 | 102 | 1.21 | 99.2 |
| SG-1st | 16 | 72 | 0.361 | 98.9 |
| SG-2nd | 14 | 70 | 0.321 | 97.5 |
| SG-3rd | 14 | 70 | 0.334 | 98.7 |
| SG-4th | 12 | 72 | 0.321 | 98.6 |
| SG-5th | 10 | 72 | 0.343 | 97.6 |
| AC-1st | 38 | 50 | 0.574 | 73.7 |
| AC-2nd | 28 | 44 | 0.465 | 83.6 |
| AC-3rd | 28 | 42 | 0.461 | 90.9 |
| AC-4th | 26 | 44 | 0.452 | 98.6 |
| AC-5th | 24 | 42 | 0.458 | 97.2 |
| tb/min | te/min | qe/(g·g-1) | Desorption efficiency/% | |
|---|---|---|---|---|
| 1st | 48 | 100 | 1.21 | 99.7 |
| 2nd | 46 | 98 | 1.18 | 99.5 |
| 3rd | 44 | 96 | 1.17 | 99.3 |
| 4th | 46 | 98 | 1.18 | 99.4 |
| 5th | 46 | 98 | 1.18 | 99.1 |
Table S5 Comparison of dynamic toluene adsorption parameters on MOS-10% for 5 times under dry condition
| tb/min | te/min | qe/(g·g-1) | Desorption efficiency/% | |
|---|---|---|---|---|
| 1st | 48 | 100 | 1.21 | 99.7 |
| 2nd | 46 | 98 | 1.18 | 99.5 |
| 3rd | 44 | 96 | 1.17 | 99.3 |
| 4th | 46 | 98 | 1.18 | 99.4 |
| 5th | 46 | 98 | 1.18 | 99.1 |
| Sample | SBET/(m2·g-1) | Sm/(m2·g-1) | Vt/(cm3·g-1) | Vm/(cm3·g-1) | Pore size/nm |
|---|---|---|---|---|---|
| MOS-10% | 696 | 0 | 0.887 | 0 | 2.64 |
| MOS-10%-5th | 638 | 0 | 0.830 | 0 | 2.64 |
| AC | 1654 | 652 | 1.06 | 0.48 | 5.58 |
| AC-5th | 1310 | 395 | 0.880 | 0.38 | 5.51 |
Table S6 Structural parameters of MOS-10% and AC before and after 5 times dynamic n-hexane adsorption under dry condition
| Sample | SBET/(m2·g-1) | Sm/(m2·g-1) | Vt/(cm3·g-1) | Vm/(cm3·g-1) | Pore size/nm |
|---|---|---|---|---|---|
| MOS-10% | 696 | 0 | 0.887 | 0 | 2.64 |
| MOS-10%-5th | 638 | 0 | 0.830 | 0 | 2.64 |
| AC | 1654 | 652 | 1.06 | 0.48 | 5.58 |
| AC-5th | 1310 | 395 | 0.880 | 0.38 | 5.51 |
| n-hexane | Toluene | |||||
|---|---|---|---|---|---|---|
| τ0/min | K׳/min-1 | R2 | τ0/min | K׳/min-1 | R2 | |
| 1st | 63.1 | 0.150 | 0.984 | 58.4 | 0.0948 | 0.979 |
| 2nd | 61.3 | 0.172 | 0.990 | 52.8 | 0.105 | 0.977 |
| 3rd | 56.7 | 0.174 | 0.979 | 51.5 | 0.114 | 0.987 |
| 4th | 57.1 | 0.169 | 0.986 | 54.1 | 0.111 | 0.978 |
| 5th | 58.9 | 0.165 | 0.984 | 54.2 | 0.110 | 0.982 |
Table S7 Simulation parameters of 5 times dynamic n-hexane and toluene adsorption on MOS-10% under dry condition
| n-hexane | Toluene | |||||
|---|---|---|---|---|---|---|
| τ0/min | K׳/min-1 | R2 | τ0/min | K׳/min-1 | R2 | |
| 1st | 63.1 | 0.150 | 0.984 | 58.4 | 0.0948 | 0.979 |
| 2nd | 61.3 | 0.172 | 0.990 | 52.8 | 0.105 | 0.977 |
| 3rd | 56.7 | 0.174 | 0.979 | 51.5 | 0.114 | 0.987 |
| 4th | 57.1 | 0.169 | 0.986 | 54.1 | 0.111 | 0.978 |
| 5th | 58.9 | 0.165 | 0.984 | 54.2 | 0.110 | 0.982 |
| tb/min | te/min | qe,hexane /g·g-1 | qe,t /g·g-1 adsorbent | qe,water /g·g-1 adsorbent | qe,hexane/ qe,water | Desorption efficiency /% | |
|---|---|---|---|---|---|---|---|
| 1st | 46 | 100 | 1.23 | 1.23 | 0.006 | 205 | 99.6 |
| 2nd | 44 | 98 | 1.20 | 1.20 | 0.003 | 400 | 99.7 |
| 3rd | 42 | 96 | 1.19 | 1.19 | 0.006 | 199 | 99.6 |
| 4th | 42 | 96 | 1.21 | 1.21 | 0.003 | 404 | 99.4 |
| 5th | 44 | 98 | 1.17 | 1.20 | 0.008 | 147 | 99.2 |
Table S8 Dynamic n-hexane adsorption parameters on MOS-10% for 5 times under 95% RH
| tb/min | te/min | qe,hexane /g·g-1 | qe,t /g·g-1 adsorbent | qe,water /g·g-1 adsorbent | qe,hexane/ qe,water | Desorption efficiency /% | |
|---|---|---|---|---|---|---|---|
| 1st | 46 | 100 | 1.23 | 1.23 | 0.006 | 205 | 99.6 |
| 2nd | 44 | 98 | 1.20 | 1.20 | 0.003 | 400 | 99.7 |
| 3rd | 42 | 96 | 1.19 | 1.19 | 0.006 | 199 | 99.6 |
| 4th | 42 | 96 | 1.21 | 1.21 | 0.003 | 404 | 99.4 |
| 5th | 44 | 98 | 1.17 | 1.20 | 0.008 | 147 | 99.2 |
| Dynamic adsorption capacity, qe /(g·g-1) | |||||
|---|---|---|---|---|---|
| Single component | Bi-component | ||||
| n-hexane | Toluene | n-hexane | Toluene | Total VOCs | |
| 1st | 1.23 | 1.21 | 0.642 | 0.569 | 1.21 |
| 2nd | 1.20 | 1.18 | 0.631 | 0.547 | 1.18 |
| 3rd | 1.19 | 1.17 | 0.613 | 0.532 | 1.15 |
| 4th | 1.22 | 1.18 | 0.607 | 0.550 | 1.16 |
| 5th | 1.21 | 1.18 | 0.628 | 0.547 | 1.18 |
Table S9 Comparison of simultaneous adsorption n-hexane and toluene parameters on MOS-10% for 5 times under dry condition
| Dynamic adsorption capacity, qe /(g·g-1) | |||||
|---|---|---|---|---|---|
| Single component | Bi-component | ||||
| n-hexane | Toluene | n-hexane | Toluene | Total VOCs | |
| 1st | 1.23 | 1.21 | 0.642 | 0.569 | 1.21 |
| 2nd | 1.20 | 1.18 | 0.631 | 0.547 | 1.18 |
| 3rd | 1.19 | 1.17 | 0.613 | 0.532 | 1.15 |
| 4th | 1.22 | 1.18 | 0.607 | 0.550 | 1.16 |
| 5th | 1.21 | 1.18 | 0.628 | 0.547 | 1.18 |
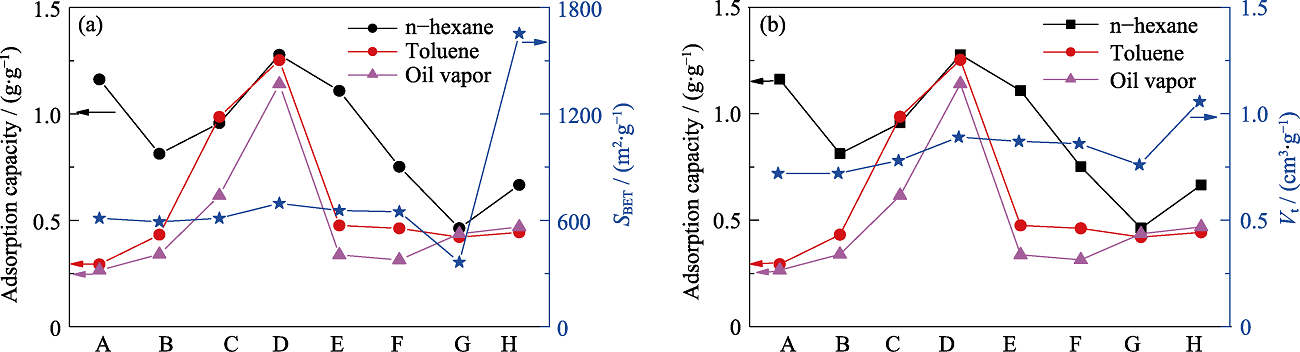
Fig. S2 Relationship between VOCs adsorption capacities and structure parameters of different samples (A) MOS-0; (B) MOS-5%; (C) MOS-7.5%; (D) MOS-10%; (E) MOS- 12.5%; (F) MOS-15%; (G) SG; (H) AC
| [1] |
ZHAO X S, MA Q, LU G Q M. VOC removal: comparison of MCM-41 with hydrophobic zeolites and activated carbon. Energ. Fuel, 1998, 12(6): 1051-1054.
DOI URL |
| [2] | ZHANG G, FEIZBAKHSHAN M, ZHENG S, et al. Effects of properties of minerals adsorbents for the adsorption and desorption of volatile organic compounds (VOC). Appl. Clay Sci., 2019, 173: 88-96. |
| [3] | TRAN THANH T, MANH TRUNG T, FELLER J F, et al. Graphene and metal organic frameworks (MOFs) hybridization for tunable chemoresistive sensors for detection of volatile organic compounds (VOCs) biomarkers. Carbon, 2020, 162: 662-662. |
| [4] |
LIU S, PENG Y, YAN T, et al. Modified silica adsorbents for toluene adsorption under dry and humid conditions: impacts of pore size and surface chemistry. Langmuir, 2019, 35(27): 8927-8934.
DOI URL |
| [5] |
WANG X, HE Y, LIU C, et al. A controllable asymmetrical/ symmetrical coating strategy for architectural mesoporous organosilica nanostructures. Nanoscale, 2016, 8(28): 13581-13588.
DOI URL |
| [6] |
SUN Y, CHEN M, WU L. Controllable synthesis of hollow periodic mesoporous organosilica spheres with radial mesochannels and their degradable behavior. J. Mater. Chem. A, 2018, 6(26): 12323-12333.
DOI URL |
| [7] |
BATONNEAU-GENER I, YONLI A, TROUVE A, et al. Tailoring the hydrophobic character of mesoporous silica by silylation for VOC removal. Sep. Sci. Technol., 2010, 45(6): 768-775.
DOI URL |
| [8] |
DOU B, HU Q, LI J, et al. Adsorption performance of VOCs in ordered mesoporous silicas with different pore structures and surface chemistry. J. Hazard. Mater., 2011, 186(2/3): 1615-1624.
DOI URL |
| [9] |
ZHANG W, QU Z, LI X, et al. Comparison of dynamic adsorption/ desorption characteristics of toluene on different porous materials. J. Environ. Sci., 2012, 24(3): 520-528.
DOI URL |
| [10] |
HARTMANN M, BISCHOF C. Mechanical stability of mesoporous molecular sieve MCM-48 studied by adsorption of benzene, n-heptane, and cyclohexane. J. Phys. Chem. B, 1999, 103(30): 6230-6235.
DOI URL |
| [11] | YANG K, SUN Q, XUE F, et al. Adsorption of volatile organic compounds by metal-organic frameworks MIL-101: Influence of molecular size and shape. J. Hazard. Mater., 2011, 195: 124-131. |
| [12] |
HU Q, LI J J, HAO Z P, et al. Dynamic adsorption of volatile organic compounds on organofunctionalized SBA-15 materials. Chem. Eng. J., 2009, 149(1/2/3): 281-288.
DOI URL |
| [13] |
KUBO S, KOSUGE K. Salt-induced formation of uniform fiberlike SBA-15 mesoporous silica particles and application to toluene adsorption. Langmuir, 2007, 23(23): 11761-11768.
DOI URL |
| [14] | QIN Y, WANG Y, WANG H, et al. Effect of morphology and pore structure of SBA-15 on toluene dynamic adsorption/desorption performance. 4th International Symposium on Environmental Science and Technology (ISEST), Dalian, 2013:366-371. |
| [15] | LIU S, CHEN J, PENG Y, et al. Studies on toluene adsorption performance and hydrophobic property in phenyl functionalized KIT-6. Chem. Eng. J., 2018, 334: 191-197. |
| [16] |
DOU B, LI J, HU Q, et al. Hydrophobic micro/mesoporous silica spheres assembled from zeolite precursors in acidic media for aromatics adsorption. Micropor. Mesopor. Mat., 2010, 133(1/2/3): 115-123.
DOI URL |
| [17] |
WANG H, TANG M, HAN L, et al. Synthesis of hollow organosiliceous spheres for volatile organic compound removal. J. Mater. Chem. A, 2014, 2(45): 19298-19307.
DOI URL |
| [18] | WANG H, TANG M, ZHANG K, et al. Functionalized hollow siliceous spheres for VOCs removal with high efficiency and stability. J. Hazard. Mater., 2014, 268: 115-123. |
| [19] |
WANG H, RONG X, HAN L, et al. Controlled synthesis of hexagonal mesostructure silica and macroporous ordered siliceous foams for VOCs adsorption. RSC Adv., 2015, 5(8): 5695-5703.
DOI URL |
| [20] | WANG J, FENG S, SONG Y, et al. Synthesis of hierarchically porous carbon spheres with yolk-shell structure for high performance supercapacitors. Catal. Today, 2015, 243: 199-208. |
| [21] | ZHANG C, WU C, HAN W, et al. Controllable synthesis of multi-morphological hollow mesoporous SiO2 and adsorption reduction of Cu2+ by its composites. Chem. J. Chinese U., 2019, 40(11): 2412-2418. |
| [22] | 王小文, 胡芸, 黄晶. 等. 疏水性分子筛对焦化废水生物处理尾水的吸附过程解析. 环境科学学报, 2012, 3(2): 2058-2065. |
| [23] |
LIU W, MA N, LI S, et al. A one-step method for pore expansion and enlargement of hollow cavity of hollow periodic mesoporous organosilica spheres. J. Mater. Sci., 2017, 52(5): 2868-2878.
DOI URL |
| [24] |
GAO M, HAN S, HU Y, et al. A pH-driven molecular shuttle based on rotaxane-bridged periodic mesoporous organosilicas with responsive release of guests. RSC Adv., 2016, 6(33): 27922-27932.
DOI URL |
| [25] | MATYSIAK W, TANSKI T. Analysis of the morphology, structure and optical properties of 1D SiO2 nanostructures obtained with Sol-Gel and electrospinning methods. Appl. Surf. Sci., 2019, 489: 34-43. |
| [26] |
CHEN J, SUN C, HUANG Z, et al. Fabrication of functionalized porous silica nanocapsules with a hollow structure for high performance of toluene adsorption-desorption. ACS Omega, 2020, 5(11): 5805-5814.
DOI URL |
| [27] | YUAN W W, YUAN P, LIU D, et al. A hierarchically porous diatomite/silicalite-1 composite for benzene adsorption/desorption fabricated via a facile pre-modification in situ synthesis route. Chem. Eng. J., 2016, 294: 333-342. |
| [28] | RAJABI H, MOSLEH M H, PRAKOSO T, et al. Competitive adsorption of multicomponent volatile organic compounds on biochar. Chemosphere, 2021, 283: 131288. |
| [1] | WANG Bo, YU Jian, LI Cuncheng, NIE Xiaolei, ZHU Wanting, WEI Ping, ZHAO Wenyu, ZHANG Qingjie. Service Stability of Gd/Bi0.5Sb1.5Te3 Thermo-electro-magnetic Gradient Composites [J]. Journal of Inorganic Materials, 2023, 38(6): 663-670. |
| [2] | WANG Shiyi, FENG Aihu, LI Xiaoyan, YU Yun. Pb (II) Adsorption Process of Fe3O4 Supported Ti3C2Tx [J]. Journal of Inorganic Materials, 2023, 38(5): 521-528. |
| [3] | LI Yue, ZHANG Xuliang, JING Fangli, HU Zhanggui, WU Yicheng. Growth and Property of Ce3+-doped La2CaB10O19 Crystal [J]. Journal of Inorganic Materials, 2023, 38(5): 583-588. |
| [4] | LUO Shilin, ZHANG Shengtai, XU Baoliang, WANG Lingkun, DUAN Siyihan, DING Yi, ZHAO Qian, DUAN Tao. Immobilizing Behavior of Trivalent Actinide Nuclides by YIG Ceramics [J]. Journal of Inorganic Materials, 2022, 37(7): 757-763. |
| [5] | JIANG Yiyi, SHEN Min, SONG Banxia, LI Nan, DING Xianghuan, GUO Leyi, MA Guoqiang. Effect of Dual-functional Electrolyte Additive on High Temperature and High Voltage Performance of Li-ion Battery [J]. Journal of Inorganic Materials, 2022, 37(7): 710-716. |
| [6] | LIU Dingwei, ZENG Jiangtao, ZHENG Liaoying, MAN Zhenyong, RUAN Xuezheng, SHI Xue, LI Guorong. High Piezoelectric Property and Low Electric Field-strain Hysteresis of BiAlO3-doped PZT Ceramics [J]. Journal of Inorganic Materials, 2022, 37(12): 1365-1370. |
| [7] | YANG Xinyue, DONG Qingshun, ZHAO Weidong, SHI Yantao. 4-Chlorobenzylamine-based 2D/3D Perovskite Solar Cells [J]. Journal of Inorganic Materials, 2022, 37(1): 72-78. |
| [8] | YU Xiangkun, LIU Kun, LI Zhipeng, ZHAO Yulu, SHEN Jinyou, MAO Ping, SUN Aiwu, JIANG Jinlong. Efficient Adsorption of Radioactive Iodide by Copper/Palygorskite Composite [J]. Journal of Inorganic Materials, 2021, 36(8): 856-864. |
| [9] | GUO Meng, ZHANG Fengnian, MIAO Yang, LIU Yufeng, YU Jun, GAO Feng. Preparation and Electrical Properties of High Entropy La(Co0.2Cr0.2Fe0.2Mn0.2Ni0.2)O3 Perovskite Ceramics Powder [J]. Journal of Inorganic Materials, 2021, 36(4): 431-435. |
| [10] | ZENG Fanxin, LIU Chuang, CAO Yuliang. Sodium Storage Behavior of Nanoporous Sb/MCNT Anode Material with High Cycle Stability by Dealloying Route [J]. Journal of Inorganic Materials, 2021, 36(11): 1137-1144. |
| [11] | DUAN Tao, DING Yi, LUO Shilin, ZHANG Shengtai, LIU Jian. Radionuclides from Nature to Nature: Recent Progress in Immobilization of High Level Nuclear Wastes in SYNROC [J]. Journal of Inorganic Materials, 2021, 36(1): 25-35. |
| [12] | ZHANG Zhigang,LU Xiaotong,LIU Jinli. NiFe2O4 Ceramic U-shaped Sleeve Prepared by Slip Casting and Pressureless Sintering [J]. Journal of Inorganic Materials, 2020, 35(6): 661-668. |
| [13] | CHEN Bowen, WANG Jingxiao, JIANG Youlin, ZHOU Haijun, LIAO Chunjing, ZHANG Xiangyu, KAN Yanmei, NI Dewei, DONG Shaoming. Stable Zirconium Carbide Fibers Fabricated by Centrifugal Spinning Technique [J]. Journal of Inorganic Materials, 2020, 35(12): 1385-1390. |
| [14] | YANG Dandan, LI Xiaoming, MENG Cuifang, CHEN Jiaxin, ZENG Haibo. Research Progress on the Stability of CsPbX3 Nanocrystals [J]. Journal of Inorganic Materials, 2020, 35(10): 1088-1098. |
| [15] | QI Xin-Xin, SONG Guang-Ping, YIN Wei-Long, WANG Ming-Fu, HE Xiao-Dong, ZHENG Yong-Ting, WANG Rong-Guo, BAI Yue-Lei. Analysis on Phase Stability and Mechanical Property of Newly-discovered Ternary Layered Boride Cr4AlB4 [J]. Journal of Inorganic Materials, 2020, 35(1): 53-60. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||