
Journal of Inorganic Materials ›› 2020, Vol. 35 ›› Issue (3): 381-389.DOI: 10.15541/jim20190365
Special Issue: 2020年环境材料论文精选(一)放射性元素去除; 2019~2020年度优秀作者作品欣赏:环境材料; 【虚拟专辑】放射性污染物去除(2020~2021)
Previous Articles Next Articles
PANG Hongwei1,TANG Hao1,WANG Jiaqi2,WANG Xiangxue1,2,YU Shujun1( )
)
Received:2019-07-19
Revised:2019-08-24
Published:2020-03-20
Online:2019-10-23
About author:PANG Hongwei(1994-), male, PhD candidate. E-mail: panghw1994@163.com
Supported by:CLC Number:
PANG Hongwei, TANG Hao, WANG Jiaqi, WANG Xiangxue, YU Shujun. Ternary Layered Double Hydroxide Supported Sulfide NZVI: Efficient U(VI) Elimination and Mechanism[J]. Journal of Inorganic Materials, 2020, 35(3): 381-389.
| Material | Pseudo-first-order model | Pseudo-second-order model | ||||
|---|---|---|---|---|---|---|
| k1/min-1 | Qe/(mg·g-1) | R2 | k2/(g·mg-1·min-1) | Qe/(mg·g-1) | R2 | |
| CMAL | 0.049 | 62.62 | 0.95 | 0.0015 | 65.36 | 0.99 |
| CMAL-SNZVI | 0.121 | 87.60 | 0.94 | 0.0036 | 90.09 | 0.99 |
Table 1 Kinetic modeling of U(VI) adsorption on CMAL and CMAL-SNZVI
| Material | Pseudo-first-order model | Pseudo-second-order model | ||||
|---|---|---|---|---|---|---|
| k1/min-1 | Qe/(mg·g-1) | R2 | k2/(g·mg-1·min-1) | Qe/(mg·g-1) | R2 | |
| CMAL | 0.049 | 62.62 | 0.95 | 0.0015 | 65.36 | 0.99 |
| CMAL-SNZVI | 0.121 | 87.60 | 0.94 | 0.0036 | 90.09 | 0.99 |
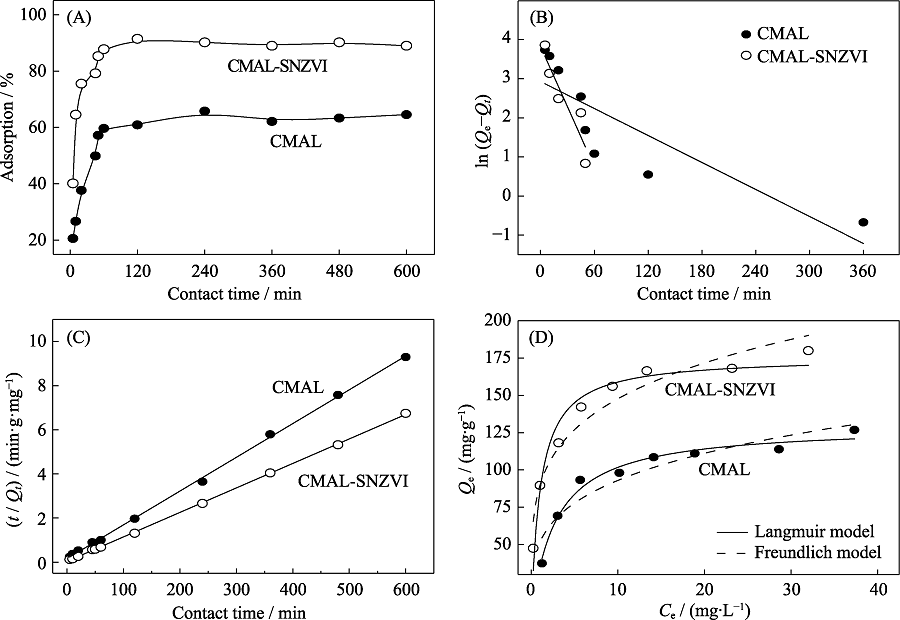
Fig. 4 Adsorption kinetics (A), pseudo-first-order (B) and pseudo-second-order kinetic plots (C), adsorption isotherms (D) of U(VI) adsorption on CMAL-SNZVI and CMAL
| Adsorbents | Langmuir model | Freundlich model | ||||
|---|---|---|---|---|---|---|
| Qmax/(mg·g-1) | KL/(L·mg-1) | R2 | KF/(mg1-n·Ln·g-1) | n | R2 | |
| CMAL | 129.8 | 0.36 | 0.975 | 51.5 | 3.89 | 0.877 |
| CMAL | 175.7 | 0.95 | 0.954 | 89.5 | 4.60 | 0.933 |
| -SNZVI | ||||||
Table 2 Parameters calculated from the Langmuir and Freundlich models for U(VI) adsorption on CMAL-SNZVI at 25 ℃
| Adsorbents | Langmuir model | Freundlich model | ||||
|---|---|---|---|---|---|---|
| Qmax/(mg·g-1) | KL/(L·mg-1) | R2 | KF/(mg1-n·Ln·g-1) | n | R2 | |
| CMAL | 129.8 | 0.36 | 0.975 | 51.5 | 3.89 | 0.877 |
| CMAL | 175.7 | 0.95 | 0.954 | 89.5 | 4.60 | 0.933 |
| -SNZVI | ||||||
| Adsorbents | pH | Equilibrium time/h | Removal capacity /(mg·g-1) | Ref. |
|---|---|---|---|---|
| nZVI/C composite | 4.0 | Not given | 103.1 | [25] |
| g-C3N4@Ni-Mg-Al-LDH | 5.0 | 6 | 99.7 | [26] |
| Ca-Mg-Al-LDH | 5.0 | 24 | 132.5 | [27] |
| GO@LDH | 4.5 | 10 | 159.7 | [28] |
| Magnetic biochar | 3.0 | 12 | 54.4 | [29] |
| CMAL | 5.0 | 2 | 129.8 | This study |
| CMAL-SNZVI | 5.0 | 2 | 175.7 |
Table 3 Comparison of CMAL-SNZVI with other typical adsorbents for U(VI) decontamination
| Adsorbents | pH | Equilibrium time/h | Removal capacity /(mg·g-1) | Ref. |
|---|---|---|---|---|
| nZVI/C composite | 4.0 | Not given | 103.1 | [25] |
| g-C3N4@Ni-Mg-Al-LDH | 5.0 | 6 | 99.7 | [26] |
| Ca-Mg-Al-LDH | 5.0 | 24 | 132.5 | [27] |
| GO@LDH | 4.5 | 10 | 159.7 | [28] |
| Magnetic biochar | 3.0 | 12 | 54.4 | [29] |
| CMAL | 5.0 | 2 | 129.8 | This study |
| CMAL-SNZVI | 5.0 | 2 | 175.7 |
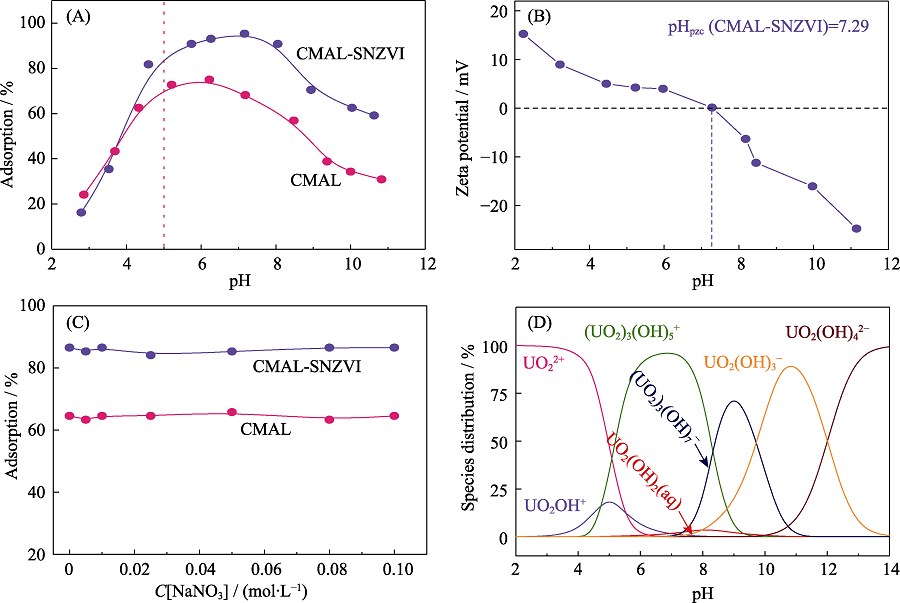
Fig. 5 Effect of pH (A) and ionic strength (C) on U(VI) adsorption of CMAL-SNZVI and CMAL, Zeta potential values of CMAL-SZVI as a function of pH (B), species distribution of U(VI) as a function of pH by Visual MINTEQ (D)
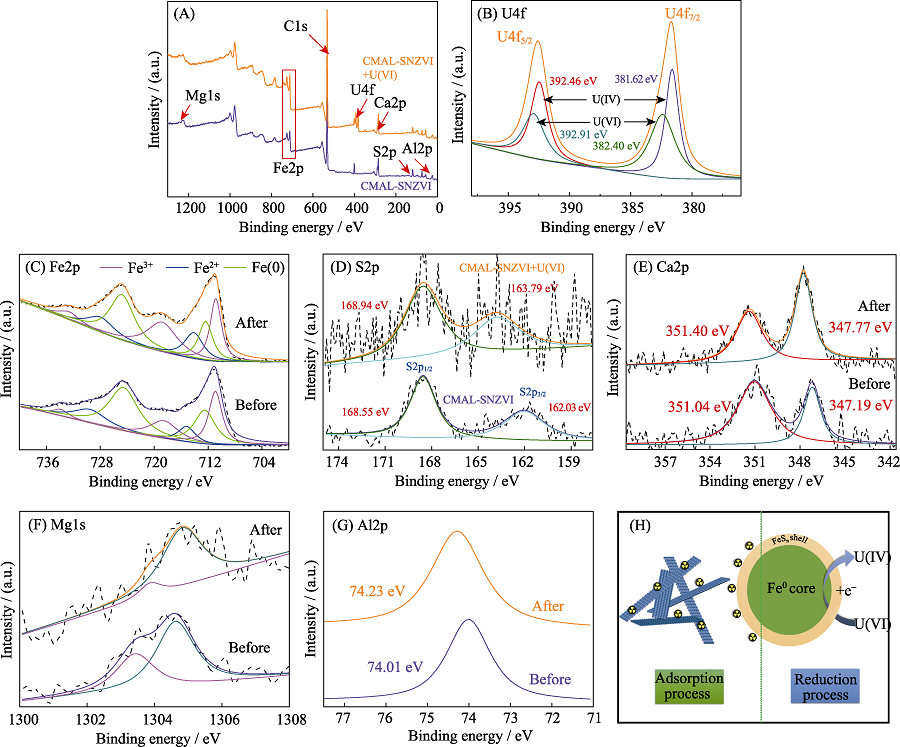
Fig. 6 XPS survey spectra of CMAL-SNZVI (before and after U(VI) adsorption) (A), High -resolution XPS spectra of U4f (B), Fe2p (C), S2p (D), Ca2p (E), Mg1s (F) and Al2p (G), and removal mechanisms of U(VI) on CMAL-SNZVI (H)
| [1] | HUANG SHU-YI, PANG HONG-WEI, LI LEI , et al. Unexpected ultrafast and high adsorption of U(VI) and Eu(III) from solution using porous Al2O3 microspheres derived from MIL-53. Chem. Eng. J., 2018,353:157-166. |
| [2] | WANG XIANG-XUE, YU SHU-QI, WU YI-HAN , et al. The synergistic elimination of uranium (VI) species from aqueous solution using bi-functional nanocomposite of carbon sphere and layered double hydroxide. Chem. Eng. J., 2018,342:321-330. |
| [3] | YU SHU-JUN, YIN LING, PANG HONG-WEI , et al. Constructing sphere-like cobalt-molybdenum-nickel ternary hydroxide and calcined ternary oxide nanocomposites for efficient removal of U(VI) from aqueous solutions. Chem. Eng. J., 2018,352:360-370. |
| [4] | GU PENG-CHENG, ZHANG SAI, LI XING , et al. Recent advances in layered double hydroxide-based nanomaterials for the removal of radionuclides from aqueous solution. Environ. Pollut., 2018,240:493-505. |
| [5] | LI JIE, WANG XIANG-XUE, ZHAO GUI-XIA , et al. Metal- organic framework-based materials: superior adsorbents for the capture of toxic and radioactive metal ions. Chem. Soc. Rev., 2018,47(7):2322-2356. |
| [6] | LIU YUE, WU YI-HAN, PANG HONG-WEI , et al. Study on the removal of water pollutants by graphite phase carbon nitride materials. Prog. Chem., 2019,31(6):831-846. |
| [7] | HU BAO-WEI, MEI XUE, LI XUE , et al. Decontamination of U(VI) from nZVI/CNF composites investigated by batch, spectroscopic and modeling techniques. J. Mol. Liq., 2017,237:1-9. |
| [8] | SHENG GUO-DONG, TANG YAN-NA, LINGHU WEN-SHENG , et al. Enhanced immobilization of ReO4- by nanoscale zerovalent iron supported on layered double hydroxide via an advanced XAFS approach: implications for TcO4- sequestration. Appl. Catal. B-Environ., 2016,192:268-276. |
| [9] | SU YI-MING, JASSBY DAVID, SONG SHIKUN , et al. Enhanced oxidative and adsorptive removal of diclofenac in heterogeneous fenton-like reaction with sulfide modified nanoscale zerovalent iron. Environ. Sci. Technol., 2018,52(11):6466-6475. |
| [10] | WU YI-HAN, PANG HONG-WEI, LIU YUE , et al. Environmental remediation of heavy metal ions by novel-nanomaterials: a review. Environ. Pollut., 2019,246:608-620. |
| [11] | ZOU YI-DONG, WANG XIANG-XUE, KHAN AYUB , et al.Environmental remediation and application of nanoscale zero-valent iron and its composites for the removal of heavy metal ions: a review. Environ. Sci. Technol., 2016,50(14):7290-7304. |
| [12] | CHEN HAI-JUN, HUANG SHU-YI, ZHANG ZHI-BIN , et al. Synthesis of functional nanoscale zero-valent iron composites for the application of radioactive uranium enrichment from environment: a review. Prog. Chem., 2017,75(6):560-574. |
| [13] | XU CONG-BIN, YANG WEN-JIE, SUN HONG-LIANG , et al. Performance and mechanism of Pb(II) removal by expanded graphite loaded with zero-valent iron. J. Inorg. Mater., 2018,33(1):41-47. |
| [14] | LI JIN-XIANG, ZHANG XUE-YING, SUN YUAN-KUI , et al. Advances in sulfidation of zerovalent iron for water decontamination. Environ. Sci. Technol., 2017,51(23):13533-13544. |
| [15] | CAO ZHEN, LIU XUE, XU JIANG , et al. Removal of antibiotic florfenicol by sulfide-modified nanoscale zero-valent iron. Environ. Sci. Technol., 2017,51(19):11269-11277. |
| [16] | HUANG SHU-YI, SONG SHUANG, ZHANG RUI , et al. Construction of layered double hydroxides/hollow carbon microsphere composites and its applications for mutual removal of Pb(II) and humic acid from aqueous solutions. ACS Sustain. Chem. Eng., 2017,5(12):11268-11279. |
| [17] | PANG HONG-WEI, WANG XIANG-XUE, YAO WEN , et al. Removal of radionuclides by metal oxide materials and mechanism research. Sci. Sin. Chim., 2018,48(1):58-73. |
| [18] | WANG XIANG-XUE, CHEN LONG, WANG LIN , et al. Synthesis of novel nanomaterials and their application in efficient removal of radionuclides. Sci. China Chem., 2019,62(8):933-967. |
| [19] | WU YI-HAN, PANG HONG-WEI, YAO WEN , et al. Synthesis of rod-like metal-organic framework (MOF-5) nanomaterial for efficient removal of U(VI): batch experiments and spectroscopy study. Sci. Bull., 2018,63(13):831-839. |
| [20] | PANG HONG-WEI, DIAO ZHUO-FAN, WANG XIANG-XUE , et al. Adsorptive and reductive removal of U(VI) by dictyophora indusiate-derived biochar supported sulfide NZVI from wastewater. Chem. Eng. J., 2019,366:368-377. |
| [21] | PANG HONG-WEI, WU YI-HAN, HUANG SHU-YI , et al. Macroscopic and microscopic investigation of uranium elimination by Ca-Mg-Al-layered double hydroxide supported nanoscale zero valent iron. Inorg. Chem. Front., 2018,5(10):2657-2665. |
| [22] | JIN QING-QING, ZHANG SAI, WEN TAO , et al. Simultaneous adsorption and oxidative degradation of bisphenol A by zero- valent iron/iron carbide nanoparticles encapsulated in N-doped carbon matrix. Environ. Pollut, 2018,243:218-227. |
| [23] | YU SHU-JUN, LIU YANG, AI YUE-JIE , et al. Rational design of carbonaceous nanofiber/Ni-Al layered double hydroxide nanocomposites for high-efficiency removal of heavy metals from aqueous solutions. Environ. Pollut., 2018,242:1-11. |
| [24] | YU SHU-JUN, WANG XIANG-XUE, CHEN ZHONG-SHAN , et al. Layered double hydroxide intercalated with aromatic acid anions for the efficient capture of aniline from aqueous solution. J. Hazard. Mater., 2016,321:111-120. |
| [25] | GU PENG-CHENG, XING JIN-LU, WEN TAO , et al. Experimental and theoretical calculation investigation on efficient Pb(II) adsorption on etched Ti3AlC2 nanofibers and nanosheets. Environ. Sci: Nano., 2018,5(4):946-955. |
| [26] | ZOU YI-DONG, LIU YANG, WANG XIANG-XUE , et al. Glycerol- modified binary layered double hydroxide nanocomposites for uranium immobilization via extended X-ray absorption fine structure technique and density functional theory calculation. ACS Sustain. Chem. Eng., 2017,5(4):3583-3595. |
| [27] | ZOU YI-DONG, WANG XIANG-XUE, WU FEN , et al. Controllable synthesis of Ca-Mg-Al layered double hydroxides and calcined layered double oxides for the efficient removal of U(VI) from wastewater solutions. ACS Sustain. Chem. Eng., 2017,5(1):1173-1185. |
| [28] | YU SHU-JUN, WANG JIAN, SONG SHUANG , et al. One-pot synthesis of graphene oxide and Ni-Al layered double hydroxides nanocomposites for the efficient removal of U(VI) from wastewater. Sci. China Chem., 2017,60(3):415-422. |
| [29] | HU QING-YUAN, ZHU YU-LING, HU BAO-WEI , et al. Mechanistic insights into sequestration of U(VI) toward magnetic biochar: batch, XPS and EXAFS techniques. J. Environ. Sci., 2018,70:217-225. |
| [30] | BHATTACHARJEE SOURJYA, GHOSHAL SUBHASIS . Optimal design of sulfidated nanoscale zerovalent iron for enhanced trichloroethene degradation. Environ. Sci. Technol., 2018,52(19):11078-11086. |
| [31] | SHENG GUO-DONG, ALSAEDI AHMED, SHAMMAKH WAFA , et al. Enhanced sequestration of selenite in water by nanoscale zero valent iron immobilization on carbon nanotubes by a combined batch, XPS and XAFS investigation. Carbon, 2016,99:123-130. |
| [32] | FAN JIA-JIA, HU YI-BO, LI XIAO-YAN . Nanoscale zero-valent iron coated with magnesium hydroxide for effective removal of cyanobacteria from water. ACS Sustain. Chem. Eng., 2018,6(11):15135-15142. |
| [33] | XU JIANG, WANG YAN, WENG CINDY , et al. Reactivity, selectivity, and long-term performance of sulfidized nanoscale zerovalent iron with different properties. Environ. Sci. Technol., 2019,53(10):5936-5945. |
| [1] | MA Xiaosen, ZHANG Lichen, LIU Yanchao, WANG Quanhua, ZHENG Jiajun, LI Ruifeng. 13X@SiO2: Synthesis and Toluene Adsorption [J]. Journal of Inorganic Materials, 2023, 38(5): 537-543. |
| [2] | GUO Chunxia, CHEN Weidong, YAN Shufang, ZHAO Xueping, YANG Ao, MA Wen. Adsorption of Arsenate in Water by Zirconia-halloysite Nanotube Material [J]. Journal of Inorganic Materials, 2023, 38(5): 529-536. |
| [3] | WANG Shiyi, FENG Aihu, LI Xiaoyan, YU Yun. Pb (II) Adsorption Process of Fe3O4 Supported Ti3C2Tx [J]. Journal of Inorganic Materials, 2023, 38(5): 521-528. |
| [4] | YU Yefan, XU Ling, NI Zhongbing, SHI Dongjian, CHEN Mingqing. Prussian Blue Modified Biochar: Preparation and Adsorption of Ammonia Nitrogen from Sewage [J]. Journal of Inorganic Materials, 2023, 38(2): 205-212. |
| [5] | WANG Hongning, HUANG Li, QING Jiang, MA Tengzhou, HUANG Weiqiu, CHEN Ruoyu. Mesoporous Organic-inorganic Hybrid Siliceous Hollow Spheres: Synthesis and VOCs Adsorption [J]. Journal of Inorganic Materials, 2022, 37(9): 991-1000. |
| [6] | HONG Jiahui, MA Ran, WU Yunchao, WEN Tao, AI Yuejie. CoNx/g-C3N4 Nanomaterials Preparation by MOFs Self-sacrificing Template Method for Efficient Photocatalytic Reduction of U(VI) [J]. Journal of Inorganic Materials, 2022, 37(7): 741-749. |
| [7] | SUN Lian, GU Quanchao, YANG Yaping, WANG Honglei, YU Jinshan, ZHOU Xingui. Two-dimensional Transition Metal Dichalcogenides for Electrocatalytic Oxygen Reduction Reaction [J]. Journal of Inorganic Materials, 2022, 37(7): 697-709. |
| [8] | MA Hui, TAO Jianghui, WANG Yanni, HAN Yu, WANG Yabin, DING Xiuping. Gold Nanoparticles Supported on Silica & Titania Hybrid Mesoporous Spheres and Their Catalytic Performance Regulation [J]. Journal of Inorganic Materials, 2022, 37(4): 404-412. |
| [9] | JIANG Lili, XU Shuaishuai, XIA Baokai, CHEN Sheng, ZHU Junwu. Defect Engineering of Graphene Hybrid Catalysts for Oxygen Reduction Reactions [J]. Journal of Inorganic Materials, 2022, 37(2): 215-222. |
| [10] | LI Chengjin, XUE Yi, ZHOU Xiaoxia, CHEN Hangrong. BiZnx/Si Photocathode: Preparation and CO2 Reduction Performance [J]. Journal of Inorganic Materials, 2022, 37(10): 1093-1101. |
| [11] | WU Jing, YU Libing, LIU Shuaishuai, HUANG Qiuyan, JIANG Shanshan, ANTON Matveev, WANG Lianli, SONG Erhong, XIAO Beibei. NiN4/Cr Embedded Graphene for Electrochemical Nitrogen Fixation [J]. Journal of Inorganic Materials, 2022, 37(10): 1141-1148. |
| [12] | LIU Cheng, ZHAO Qian, MOU Zhiwei, LEI Jiehong, DUAN Tao. Adsorption Properties of Novel Bismuth-based SiOCNF Composite Membrane for Radioactive Gaseous Iodine [J]. Journal of Inorganic Materials, 2022, 37(10): 1043-1050. |
| [13] | GAO Wa, XIONG Yujie, WU Congping, ZHOU Yong, ZOU Zhigang. Recent Progress on Photocatalytic CO2 Reduction with Ultrathin Nanostructures [J]. Journal of Inorganic Materials, 2022, 37(1): 3-14. |
| [14] | LIU Peng, WU Shimiao, WU Yunfeng, ZHANG Ning. Synthesis of Zn0.4(CuGa)0.3Ga2S4/CdS Photocatalyst for CO2 Reduction [J]. Journal of Inorganic Materials, 2022, 37(1): 15-21. |
| [15] | GUO Lina, HE Xuebing, LYU Lin, WU Dan, YUAN Hong. Modulation of CuO Surface Properties for Selective Electrocatalytic Reduction of CO2 to HCOOH [J]. Journal of Inorganic Materials, 2022, 37(1): 29-37. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||