无机材料学报 ›› 2021, Vol. 36 ›› Issue (4): 339-346.DOI: 10.15541/jim20200611
所属专题: 【结构材料】热障与环境障涂层; 【结构材料】高熵陶瓷
孙鲁超1( ), 任孝旻1,2, 杜铁锋1, 罗颐秀1, 张洁1, 王京阳1(
), 任孝旻1,2, 杜铁锋1, 罗颐秀1, 张洁1, 王京阳1( )
)
收稿日期:2020-10-27
修回日期:2020-12-14
出版日期:2021-04-20
网络出版日期:2020-12-10
通讯作者:
王京阳, 研究员. E-mail: jywang@imr.ac.cn
作者简介:孙鲁超(1984-), 男, 副研究员. E-mail: lcsun@imr.ac.cn
基金资助:
SUN Luchao1( ), REN Xiaomin1,2, DU Tiefeng1, LUO Yixiu1, ZHANG Jie1, WANG Jingyang1(
), REN Xiaomin1,2, DU Tiefeng1, LUO Yixiu1, ZHANG Jie1, WANG Jingyang1( )
)
Received:2020-10-27
Revised:2020-12-14
Published:2021-04-20
Online:2020-12-10
Contact:
WANG Jingyang, professor. E-mail: jywang@imr.ac.cn
About author:SUN Luchao(1984-), male, associate professor. E-mail: lcsun@imr.ac.cn
Supported by:摘要:
环境障涂层是先进航空发动机高温结构部件用碳化硅纤维增强碳化硅(SiCf/SiC)陶瓷基复合材料的关键防护。稀土硅酸盐具有低热膨胀系数、优良的抗水氧/CMAS腐蚀性能以及与硅基陶瓷良好的化学相容性, 是目前国际公认的优选环境障涂层体系。常规含单一稀土元素的稀土硅酸盐环境障涂层材料, 存在热应力失配、高温相转变和耐腐蚀性能差等问题, 尚无法完全满足极端燃气环境中的长寿命服役要求。本综述介绍了为解决稀土硅酸盐环境障涂层的关键性能局限, 利用高熵化合物设计方法, 针对稀土硅酸盐热学性能(热膨胀系数和热导率)调控、耐CMAS腐蚀性能提升和相结构稳定性优化方面获得的新进展。这些研究进展为稀土硅酸盐材料的创新设计提供了新思路, 为其作为下一代环境障涂层的性能突破提供了支撑。
中图分类号:
孙鲁超, 任孝旻, 杜铁锋, 罗颐秀, 张洁, 王京阳. 高熵化设计: 稀土硅酸盐材料关键性能优化新策略[J]. 无机材料学报, 2021, 36(4): 339-346.
SUN Luchao, REN Xiaomin, DU Tiefeng, LUO Yixiu, ZHANG Jie, WANG Jingyang. High Entropy Engineering: New Strategy for the Critical Property Optimizations of Rare Earth Silicates[J]. Journal of Inorganic Materials, 2021, 36(4): 339-346.
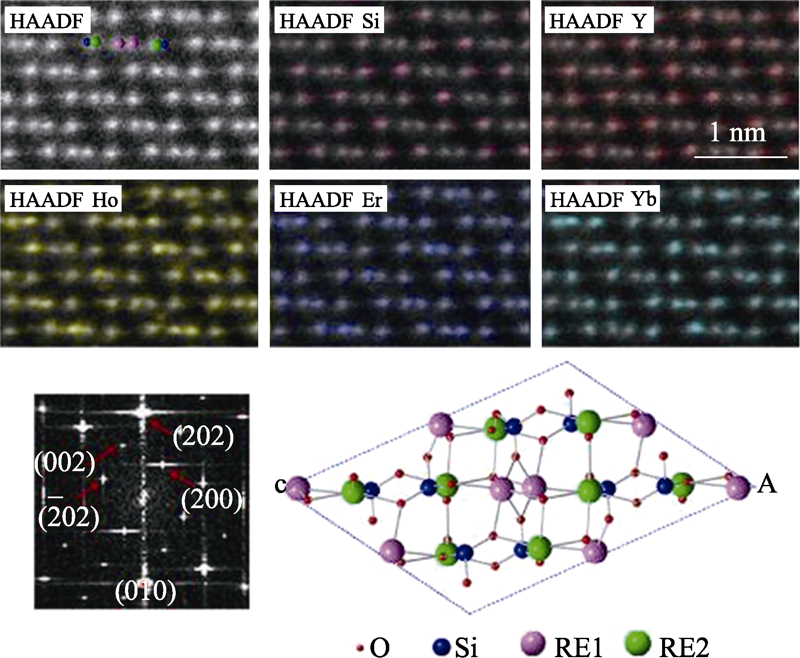
图1 高熵稀土单硅酸盐(Y1/4Ho1/4Er1/4Yb1/4)2SiO5的HAADF- STEM照片及其原子尺度元素分布图[40]
Fig. 1 HAADF-STEM image of high entropy (Y1/4Ho1/4Er1/4Yb1/4)2- SiO5, EDS mapping of uniform spatial distributions for each element[40]
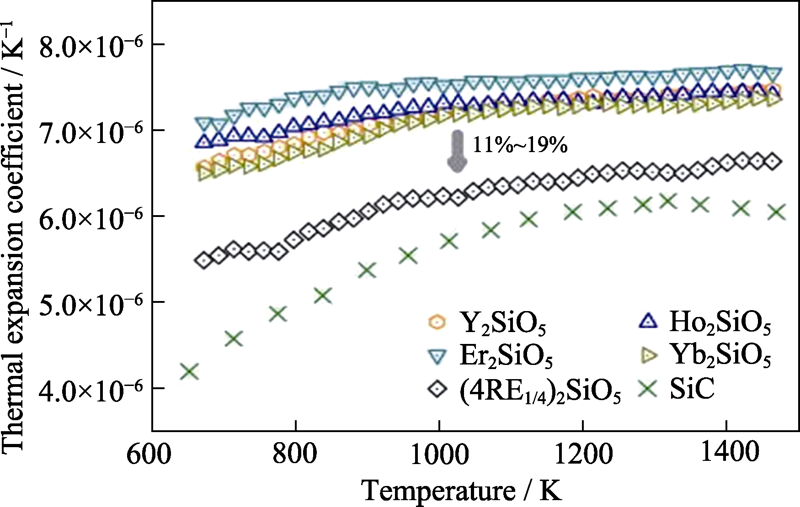
图2 高熵稀土单硅酸盐(Y1/4Ho1/4Er1/4Yb1/4)2SiO5的热膨胀系数随温度变化关系[40]
Fig. 2 Temperature dependent thermal expansion coefficient of high entropy (Y1/4Ho1/4Er1/4Yb1/4)2SiO5[40]

图3 高熵稀土双硅酸盐(Er1/4Tm1/4Yb1/4Lu1/4)2Si2O7在1500 ℃高温CMAS腐蚀(a~b)4 h和(c~d)50 h后样品表面形貌[48]
Fig. 3 Surface observations of high entropy (Er1/4Tm1/4Yb1/4Lu1/4)2Si2O7 after CMAS corrosion at 1500 ℃ for 4 h (a-b) and 50 h (c-d) [48]

图4 高熵稀土双硅酸盐(Er1/4Tm1/4Yb1/4Lu1/4)2Si2O7在1500 ℃下CMAS腐蚀(a~b)4 h和(c~d)50 h截面形貌[48]
Fig. 4 Observations of the reaction front in the cross-sections of high entropy (Er1/4Tm1/4Yb1/4Lu1/4)2Si2O7 after CMAS corrosion at 1500 ℃ for 4 h (a,b) and 50 h (c,d)[48]
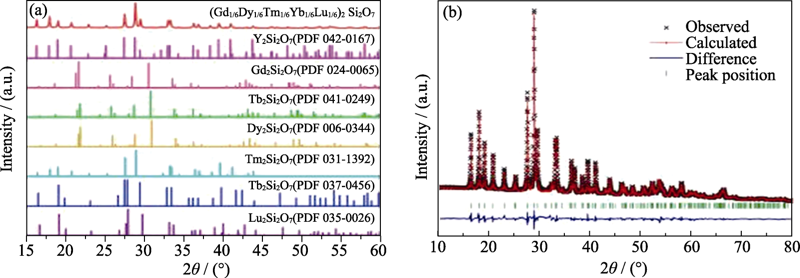
图5 (a)高熵稀土双硅酸盐(Gd1/6Tb1/6Dy1/6Tm1/6Yb1/4Lu1/6)2 Si2O7及RE2Si2O7 (RE = Y, Gd, Tb, Dy, Tm, Yb和Lu)的XRD图谱和(b)高熵稀土双硅酸盐(Gd1/6 Tb1/6Dy1/6Tm1/6Yb1/4Lu1/6)2Si2O7的 XRD图谱Rietveld精修结果[50]
Fig. 5 (a) XRD patterns of (Gd1/6Tb1/6Dy1/6Tm1/6Yb1/4Lu1/6)2 Si2O7, along with the standard XRD patterns of RE2Si2O7 (RE = Y, Gd, Tb, Dy, Tm, Yb and Lu) and (b) Rietveld refinement of XRD pattern for (Gd1/6Tb1/6Dy1/6Tm1/6Yb1/4Lu1/6)2Si2O7[50]

图6 高熵稀土双硅酸盐(Gd1/6Tb1/6Dy1/6Tm1/6Yb1/4Lu1/6)2Si2O7的(a) SEM照片和各元素分布的面扫描能谱分析; (b) HAADF-STEM及原子尺度元素分布; (c)稀土双硅酸盐全新相稳定模式示意图[50]
Fig. 6 (a) SEM image of (Gd1/6Tb1/6Dy1/6Tm1/6Yb1/4Lu1/6)2Si2O7 surface with EDS mappings of Si, Gd, Tb, Dy, Tm, Yb and Lu, (b) STEM high angle annular dark field (HAADF) image and corresponding selected compositional EDS maps of high entropy (Gd1/6Tb1/6Dy1/6Tm1/6Yb1/4Lu1/6)2Si2O7, and (c) schematic diagram of the phase formation of (6RE1/6)2Si2O7[50]
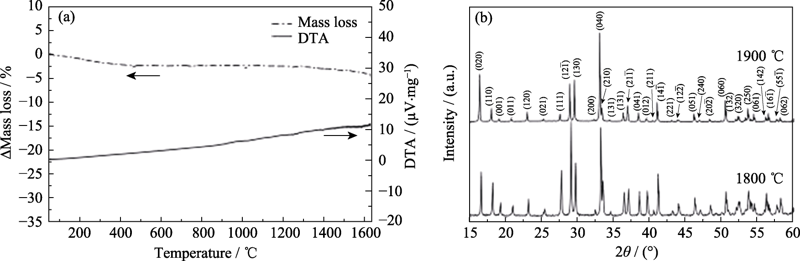
图7 高熵稀土双硅酸盐(Gd1/6Tb1/6Dy1/6Tm1/6Yb1/4Lu1/6)2 Si2O7的(a)热重-差热分析曲线和(b)1800及1900 ℃热处理2 h后样品的XRD图谱[50]
Fig. 7 (a)TG/DTA curves of (Gd1/6Tb1/6Dy1/6Tm1/6Yb1/4Lu1/6)2 Si2O7 and (b) XRD patterns of specimens after being heat-treated at 1800 and 1900 ℃ for 2 h[50]
| [1] |
TSAI M H, YEH J W. High-entropy alloys: a critical review. Materials Research Letters, 2014,2(3):107-123.
DOI URL |
| [2] |
MIRACLE D B, SENKOV O N. A critical review of high entropy alloys and related concepts. Acta Materialia, 2017,122:448-511.
DOI URL |
| [3] |
YEH J W, CHEN S K, LIN S J, et al. Nanostructured high-entropy alloys with multiple principal elements: novel alloy design concepts and outcomes. Advanced Engineering Materials, 2004,6(5):299-303.
DOI URL |
| [4] |
HUANG P K, YEH J W, SHUN T T, et al. Multi-principal-element alloys with improved oxidation and wear resistance for thermal spray coating. Advanced Engineering Materials, 2004,6(1/2):74-78.
DOI URL |
| [5] | TONG C J, CHEN Y L, CHEN S K, et al. Microstructure characterization of AlxCoCrCuFeNi high-entropy alloy system with multiprincipal elements. Metallurgical and Materials Transactions A, 2005,36(4):881-893. |
| [6] | ZHANG Y, ZUO T T, TANG Z, et al. Microstructures and properties of high-entropy alloys. Progress in Materials Science, 2014,61:1-93. |
| [7] | MURTY B S, YEH J W, RANGANATHAN S. High-entropy Alloys. London: Elsevier, 2014. |
| [8] | ZHANG Y, ZUO T T, CHENG Y Q, et al. High-entropy alloys with high saturation magnetization, electrical resistivity, and malleability. Scientific Reports, 2013,3:1-7. |
| [9] | CHUANG M H, TSAI M H, WANG W R, et al. Microstructure and wear behavior of AlxCo1.5CrFeNi1.5Tiy high entropy alloys. Acta Materialia, 2011,59(16):6308-6317. |
| [10] | JIANG S C, HU T, GILD J, et al. A new class of high-entropy perovskite oxides. Scripta Materialia, 2018,142:116-120. |
| [11] | TSAI M H. Physical properties of high entropy alloys. Entropy, 2013,15:5338-5345. |
| [12] | 顾俊峰, 邹冀, 张帆, 等. 高熵陶瓷材料研究进展. 中国材料进展, 2019,38(9):855-865. |
| [13] | YEH J W. Recent progress in high-entropy alloys. Annales De Chimie-Science des Materiaux, 2006,31:633-648. |
| [14] | MIRACLE D B. High-entropy alloys: a current evaluation of founding ideas and core effects and exploring “nonlinear alloys”. JOM, 2017,69(11):2130-2136. |
| [15] | ROST C M, SACHET E, BORMAN T, et al. Entropy-stabilized oxides. Nature Communications, 2015,6(1):8485. |
| [16] | CHELLALI M R, SARKAR A, NANDAM S H, et al. On the homogeneity of high entropy oxides: an investigation at the atomic scale. Scripta Materialia, 2019,166:58-63. |
| [17] | DJENADIC R, SARKAR A, CLEMENS O, et al. Multicomponent equiatomic rare earth oxides. Materials Research Letters, 2017,5(2):102-109. |
| [18] | DUPUY A D, WANG X, SCHOENUNG J M. Entropic phase transformation in nanocrystalline high entropy oxides. Materials Research Letters, 2019,7(2):60-67. |
| [19] |
GILD J, ZHANG Y Y, HARRINGTON T, et al. High-entropy metal diborides: a new class of high-entropy materials and a new type of ultrahigh temperature ceramics. Scientific Reports, 2016,6:37946.
URL PMID |
| [20] | YAN X L, CONSTANTIN L, LU Y F, et al. (Hf0.2Zr0.2Ta0.2Nb0.2Ti0.2)C high-entropy ceramics with low thermal conductivity. Journal of the American Ceramic Society, 2018,101(10):4486-4491. |
| [21] | CHEN H, XIANG H M, DAI F Z, et al. High porosity and low thermal conductivity high entropy (Zr0.2Hf0.2Ti0.2Nb0.2Ta0.2)C. Journal of Materials Science & Technology, 2019,35(8):1700-1705. |
| [22] | CASTLE E, CSANADI T, GRASSO S, et al. Processing and properties of high-entropy ultra-high temperature carbides. Scientific Reports, 2018,8(1):8609. |
| [23] |
SARKER P, HARRINGTON T, TOHER C, et al. High-entropy high-hardness metal carbides discovered by entropy descriptors. Nature Communications, 2018,9(1):4980.
URL PMID |
| [24] | YE B L, WEN T Q, NGUYEN M C, et al. First-principles study, fabrication and characterization of (Zr0.25Nb0.25Ti0.25V0.25)C high- entropy ceramics. Acta Materialia, 2019,170:15-23. |
| [25] | HARRINGTON T J, GILD J, SARKER P, et al. Phase stability and mechanical properties of novel high entropy transition metal carbides. Acta Materialia, 2019,166:271-280. |
| [26] | YE B L, WEN T Q, HUANG K H, et al. First-principles study, fabrication, and characterization of (Hf0.2Zr0.2Ta0.2Nb0.2Ti0.2)C high- entropy ceramic. Journal of the American Ceramic Society, 2019,102(7):4344-4352. |
| [27] | WANG K, CHEN L, XU C G, et al. Microstructure and mechanical properties of (TiZrNbTaMo)C high-entropy ceramic. Journal of Materials Science & Technology, 2020,39:99-105. |
| [28] | ZHANG W, CHEN L, XU C G, et al. Densification, microstructure and mechanical properties of multicomponent (TiZrHfNbTaMo)C ceramic prepared by pressureless sintering. Journal of Materials Science & Technology, 2021,72:23-28. |
| [29] | JIN T, SANG X H, UNOCIC R R, et al. Mechanochemical- assisted synthesis of high-entropy metal nitride via a soft urea strategy. Advanced Materials, 2018,30(23):1707512. |
| [30] |
GILD J, BRAUN J, KAUFMANN K, et al. A high-entropy silicide: (Mo0.2Nb0.2Ta0.2Ti0.2W0.2)Si2. Journal of Materiomics, 2019,5(3):337-343.
DOI URL |
| [31] | ZHAO Z F, XIANG H M, DAI F Z, et al. (TiZrHf)P2O7: an equimolar multicomponent or high entropy ceramic with good thermal stability and low thermal conductivity. Journal of Materials Science & Technology, 2019,35(10):2227-2231. |
| [32] | LIU Y C, JIA D C, ZHOU Y, et al. Zn0.1Ca0.1Sr0.4Ba0.4ZrO3: a non-equimolar multicomponent perovskite ceramic with low thermal conductivity. Journal of the European Ceramic Society, 2020,40:6272-6277. |
| [33] | ZHU D M. Advanced Environmental Barrier Coatings for SiC/SiC Ceramic Matrix Composite Turbine Components. Engineered Ceramics: Current Status and Future Prospects, Hoboken, New Jersey: John Wiley & Sons, Inc, 2016. |
| [34] | LEE K N, FOX D S, BANSAL N P. Rare earth silicate environmental barrier coatings for SiC/SiC composites and Si3N4 ceramics. Journal of the European Ceramic Society, 2005,25(10):1705-1715. |
| [35] | 田志林. 几种稀土硅酸盐陶瓷的预测、制备和性能研究. 北京: 中国科学院大学博士学位论文, 2016. |
| [36] | LUO Y X, SUN L C, WANG J M, et al. Tunable thermal properties in yttrium silicates switched by anharmonicity of low-frequency phonons. Journal of the European Ceramic Society, 2018,38:2043-2052. |
| [37] | POERSCHKE D L, HASS D D, EUSTIS S, et al. Stability and CMAS resistance of ytterbium-silicate/hafnate EBCs/TBC for SiC composites. Journal of the American Ceramic Society, 2015,98(1):278-286. |
| [38] | DONG Y, REN K, LU Y H, et al. High-entropy environmental barrier coating for the ceramic matrix composites. Journal of the European Ceramic Society, 2019,39:2574-2579. |
| [39] | CHEN H, XIANG H M, DAI F Z, et al. High entropy (Yb0.25Y0.25Lu0.25Er0.25)2SiO5 with strong anisotropy in thermal expansion. Journal of Materials Science & Technology, 2020,36:134-139. |
| [40] | REN X M, TIAN Z L, ZHANG J, et al. Equiatomic quaternary (Y1/4Ho1/4Er1/4Yb1/4)2SiO5 silicate: a perspective multifunctional thermal and environmental barrier coating material. Scripta Materialia, 2019,168:47-50. |
| [41] | RIDLEY M, GASKINS J, HOPKINS P, et al. Tailoring thermal properties of multi-component rare earth monosilicates. Acta Materialia, 2020,195:698-707. |
| [42] | TURCER L R, SENGUPTA A, PADTURE N P. Low thermal conductivity in high-entropy rare-earth pyrosilicate solid-solutions for thermal environmental barrier coatings. Scripta Materialia, 2021,191:40-45. |
| [43] | POERSCHKE D L, JACKSON R W, LEVI C G. Silicate deposit degradation of engineered coatings in gas turbines: progress toward models and materials solutions. Annual Review of Materials Research, 2017,47:297-330. |
| [44] | LIU J, ZHANG L T, LIU Q M, et al. Calcium-magnesium- aluminosilicate corrosion behaviors of rare-earth disilicates at 1400 ℃. Journal of the European Ceramic Society, 2013,33:3419-3428. |
| [45] | TIAN Z L, REN X M, LEI Y M, et al. Corrosion of RE2Si2O7 (RE=Y, Yb, and Lu) environmental barrier coating materials by molten calcium-magnesium-alumino-silicate glass at high temperatures. Journal of the European Ceramic Society, 2019,39:4245-4254. |
| [46] | TURCER L R, KRAUSE A R, GARCES H F, et al. Environmental-barrier coating ceramics for resistance against attack by molten calcia-magnesia-aluminosilicate (CMAS) glass: Part I, YAlO3 and γ-Y2Si2O7. Journal of the European Ceramic Society, 2018,38:3905-3913. |
| [47] | TURCER L R, KRAUSE A R, GARCES H F, et al. Environmental-barrier coating ceramics for resistance against attack by molten calcia-magnesia-aluminosilicate (CMAS) glass: Part II, β-Yb2Si2O7 and β-Sc2Si2O7. Journal of the European Ceramic Society, 2018,38:3914-3924. |
| [48] | SUN L C, LUO Y X, TIAN Z L, et al. High temperature corrosion of (Er0.25Tm0.25Yb0.25Lu0.25)2Si2O7 environmental barrier coating material subjected to water vapor and molten calcium-magnesium- aluminosilicate (CMAS). Corrosion Science, 2020,175:108881. |
| [49] | FELSCHE J. The Crystal Chemistry of the Rare-earth Silicates. Rare Earths. Structure and Bonding, Vol 13. Berlin, Heidelberg: Springer, 1973. |
| [50] | SUN L C, LUO Y X, REN X M, et al. A multicomponent γ-type (Gd1/6Tb1/6Dy1/6Tm1/6Yb1/6Lu1/6)2Si2O7 disilicate with outstanding thermal stability. Materials Research Letters, 2020,8(11):424-430. |
| [1] | 丁玲, 蒋瑞, 唐子龙, 杨运琼. MXene材料的纳米工程及其作为超级电容器电极材料的研究进展[J]. 无机材料学报, 2023, 38(6): 619-633. |
| [2] | 杨卓, 卢勇, 赵庆, 陈军. X射线衍射Rietveld精修及其在锂离子电池正极材料中的应用[J]. 无机材料学报, 2023, 38(6): 589-605. |
| [3] | 陈强, 白书欣, 叶益聪. 热管理用高导热碳化硅陶瓷基复合材料研究进展[J]. 无机材料学报, 2023, 38(6): 634-646. |
| [4] | 林俊良, 王占杰. 铁电超晶格的研究进展[J]. 无机材料学报, 2023, 38(6): 606-618. |
| [5] | 范栋, 钟鑫, 王亚文, 张振忠, 牛亚然, 李其连, 张乐, 郑学斌. 富铝CMAS对稀土硅酸盐环境障涂层的腐蚀行为与机制研究[J]. 无机材料学报, 2023, 38(5): 544-552. |
| [6] | 牛嘉雪, 孙思, 柳鹏飞, 张晓东, 穆晓宇. 铜基纳米酶的特性及其生物医学应用[J]. 无机材料学报, 2023, 38(5): 489-502. |
| [7] | 苑景坤, 熊书锋, 陈张伟. 聚合物前驱体转化陶瓷增材制造技术研究趋势与挑战[J]. 无机材料学报, 2023, 38(5): 477-488. |
| [8] | 杜剑宇, 葛琛. 光电人工突触研究进展[J]. 无机材料学报, 2023, 38(4): 378-386. |
| [9] | 杨洋, 崔航源, 祝影, 万昌锦, 万青. 柔性神经形态晶体管研究进展[J]. 无机材料学报, 2023, 38(4): 367-377. |
| [10] | 游钧淇, 李策, 杨栋梁, 孙林锋. 氧化物双介质层忆阻器的设计及应用[J]. 无机材料学报, 2023, 38(4): 387-398. |
| [11] | 张超逸, 唐慧丽, 李宪珂, 王庆国, 罗平, 吴锋, 张晨波, 薛艳艳, 徐军, 韩建峰, 逯占文. 新型GaN与ZnO衬底ScAlMgO4晶体的研究进展[J]. 无机材料学报, 2023, 38(3): 228-242. |
| [12] | 陈昆峰, 胡乾宇, 刘锋, 薛冬峰. 多尺度晶体材料的原位表征技术与计算模拟研究进展[J]. 无机材料学报, 2023, 38(3): 256-269. |
| [13] | 齐占国, 刘磊, 王守志, 王国栋, 俞娇仙, 王忠新, 段秀兰, 徐现刚, 张雷. GaN单晶的HVPE生长与掺杂进展[J]. 无机材料学报, 2023, 38(3): 243-255. |
| [14] | 林思琪, 李艾燃, 付晨光, 李荣斌, 金敏. Zintl相Mg3X2(X=Sb, Bi)基晶体生长及热电性能研究进展[J]. 无机材料学报, 2023, 38(3): 270-279. |
| [15] | 刘岩, 张珂颖, 李天宇, 周菠, 刘学建, 黄政仁. 陶瓷材料电场辅助连接技术研究现状及发展趋势[J]. 无机材料学报, 2023, 38(2): 113-124. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||