无机材料学报 ›› 2021, Vol. 36 ›› Issue (2): 168-174.DOI: 10.15541/jim20190650
所属专题: 能源材料论文精选(2021); 【虚拟专辑】钙钛矿材料(2020~2021); 【虚拟专辑】太阳能电池(2020~2021); 【能源环境】钙钛矿; 【能源环境】太阳能电池
王艳香( ), 高培养, 范学运, 李家科, 郭平春, 黄丽群, 孙健
), 高培养, 范学运, 李家科, 郭平春, 黄丽群, 孙健
收稿日期:2019-12-25
修回日期:2020-06-17
出版日期:2021-02-20
网络出版日期:2020-07-10
作者简介:王艳香(1972-), 女, 教授. E-mail: yxwang72@163.com
基金资助:
WANG Yanxiang( ), GAO Peiyang, FAN Xueyun, LI Jiake, GUO Pingchun, HUANG Liqun, SUN Jian
), GAO Peiyang, FAN Xueyun, LI Jiake, GUO Pingchun, HUANG Liqun, SUN Jian
Received:2019-12-25
Revised:2020-06-17
Published:2021-02-20
Online:2020-07-10
About author:WANG Yanxiang(1972-), female, professor. E-mail: yxwang72@163.com
Supported by:摘要:
电子传输层是钙钛矿太阳能电池的关键部分, 起到阻挡空穴、传输电子和减少电子空穴复合的作用。本研究采用低温溶液法制备SnO2薄膜作为钙钛矿电池的电子传输层, 研究SnO2的退火温度对电子传输层微观形貌、物理性能以及钙钛矿太阳能电池性能的影响。结果表明: 当退火温度为60、90、120和240 ℃时, SnO2薄膜表面存在较多的孔隙; 而退火温度为150、180和210 ℃时, 薄膜表面孔隙较少。在实验温度下, 制备的SnO2薄膜为四方相, FTO玻璃上涂覆SnO2薄膜后其透过率要优于空白FTO玻璃的透过率。当SnO2退火温度为180 ℃时, 薄膜的电子迁移率最高, 钙钛矿电池具有最佳的传输电阻和复合电阻, 所得电池的性能最优, 其光电转换效率为17.28%, 开路电压为1.09 V, 短路电流为20.91 mA/cm2, 填充因子为75.91%。
中图分类号:
王艳香, 高培养, 范学运, 李家科, 郭平春, 黄丽群, 孙健. SnO2退火温度对钙钛矿太阳能电池性能的影响[J]. 无机材料学报, 2021, 36(2): 168-174.
WANG Yanxiang, GAO Peiyang, FAN Xueyun, LI Jiake, GUO Pingchun, HUANG Liqun, SUN Jian. Effect of SnO2 Annealing Temperature on the Performance of Perovskite Solar Cells[J]. Journal of Inorganic Materials, 2021, 36(2): 168-174.

图1 FTO和不同退火温度制得的SnO2薄膜的FSEM照片
Fig. 1 FSEM images of FTO and SnO2 films annealed at different temperatures (a, j) Bare FTO; (b) 60 ℃; (c) 90 ℃; (d) 120 ℃; (e) 150 ℃; (f, i) 180 ℃; (g) 210 ℃; (h) 240 ℃

图 S1 不同退火温度制得的SnO2薄膜的AFM图像(20 μm×20 μm)
Fig. 8 AFM images of SnO2 films prepared at different annealing temperatures (20 μm×20 μm) (a) 60 ℃; (b) 90 ℃; (c) 120 ℃; (d) 150 ℃; (e) 180 ℃; (f) 210 ℃; (g) 240 ℃

图3 以FTO为基底不同退火温度下获得的SnO2薄膜的紫外-可见吸收光谱图(彩色曲线见电子版)
Fig. 3 UV-visible absorption spectra of SnO2 films annealed at different temperatures with FTO as substrate (Colourful curves are available on web)
| Temperature/℃ | 60 | 90 | 120 | 150 | 180 | 210 | 240 |
|---|---|---|---|---|---|---|---|
| Band gap/eV | 3.88 | 3.87 | 3.88 | 3.91 | 3.90 | 3.88 | 3.88 |
表S1 不同退火温度下SnO2薄膜的带隙
Table 1 Bandgap of SnO2 films annealed at different temperatures
| Temperature/℃ | 60 | 90 | 120 | 150 | 180 | 210 | 240 |
|---|---|---|---|---|---|---|---|
| Band gap/eV | 3.88 | 3.87 | 3.88 | 3.91 | 3.90 | 3.88 | 3.88 |
| Temperature/℃ | VOC/V | JSC/(mA∙cm-2) | FF/% | PCE/% |
|---|---|---|---|---|
| 60 | 0.87 | 18.85 | 40.75 | 6.72 |
| 90 | 0.90 | 19.15 | 55.70 | 9.60 |
| 120 | 0.97 | 19.56 | 71.67 | 13.60 |
| 150 | 1.06 | 19.65 | 74.36 | 15.48 |
| 180 | 1.09 | 20.91 | 75.91 | 17.28 |
| 210 | 1.06 | 19.27 | 73.32 | 14.91 |
| 240 | 0.99 | 19.23 | 70.75 | 13.43 |
表1 以不同退火温度制得的SnO2薄膜为ETL的PSC的光电性能参数
Table 1 Photoelectric parameters of PSCs with SnO2 ETLs annealed at different temperatures
| Temperature/℃ | VOC/V | JSC/(mA∙cm-2) | FF/% | PCE/% |
|---|---|---|---|---|
| 60 | 0.87 | 18.85 | 40.75 | 6.72 |
| 90 | 0.90 | 19.15 | 55.70 | 9.60 |
| 120 | 0.97 | 19.56 | 71.67 | 13.60 |
| 150 | 1.06 | 19.65 | 74.36 | 15.48 |
| 180 | 1.09 | 20.91 | 75.91 | 17.28 |
| 210 | 1.06 | 19.27 | 73.32 | 14.91 |
| 240 | 0.99 | 19.23 | 70.75 | 13.43 |
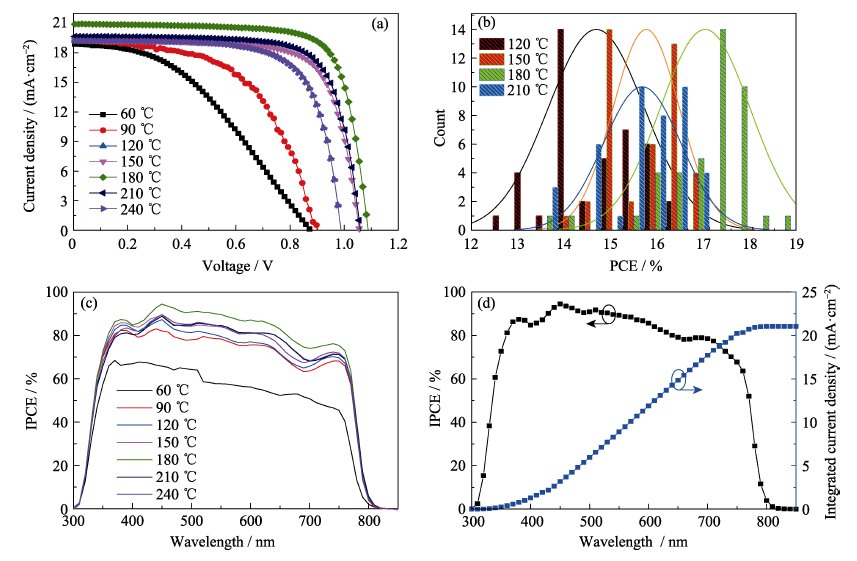
图5 以不同退火温度制得的SnO2薄膜为ETL的PSC的(a) J-V曲线, (b) PCE分布直方图, (c) IPCE曲线; (d) 以180 ℃退火温度制得的SnO2薄膜为ETL的PSC的IPCE及积分电流曲线(彩色曲线见电子版)
Fig. 5 (a) J-V curves, (b) PCE histograms, (c) IPCE spectra of PSCs prepared with SnO2 ETLs annealed at different temperatures; (d) IPCE and integral current curves of PSC with SnO2 ETL annealed at 180 ℃ (Colourful curves are available on web)
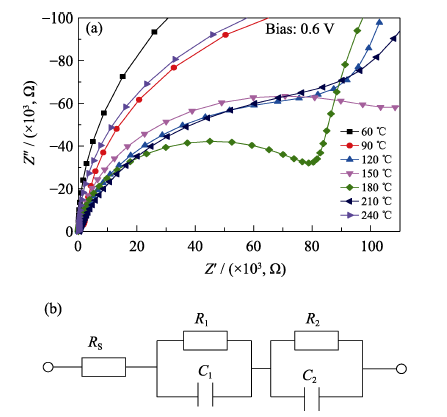
图S4 以不同退火温度制得的SnO2薄膜为ETL的PSC的(a)电化学阻抗谱局部放大图和(b)等效电路图
Fig. 11 (a) Zoom-in high-frequency region of Nyquist plot and (b) equivalent circuit for PSCs with SnO2 ETLs annealed at different temperatures
| Temperature/℃ | Resistivity/ (×10-4, Ω∙cm) | Carrier mobility/ (cm2∙V-1·s-1) |
|---|---|---|
| 60 | 1.40 | 23.11 |
| 90 | 1.74 | 25.60 |
| 120 | 1.80 | 33.98 |
| 150 | 1.24 | 45.26 |
| 180 | 1.46 | 57.89 |
| 210 | 1.64 | 39.81 |
| 240 | 1.38 | 26.21 |
表2 以FTO为基底在不同退火温度下制得的SnO2薄膜的电学性能参数
Table 2 Electrical properties of SnO2 films annealed at different temperatures with FTO as substrate
| Temperature/℃ | Resistivity/ (×10-4, Ω∙cm) | Carrier mobility/ (cm2∙V-1·s-1) |
|---|---|---|
| 60 | 1.40 | 23.11 |
| 90 | 1.74 | 25.60 |
| 120 | 1.80 | 33.98 |
| 150 | 1.24 | 45.26 |
| 180 | 1.46 | 57.89 |
| 210 | 1.64 | 39.81 |
| 240 | 1.38 | 26.21 |
| [1] | KOJIMA A, TESHIMA K, SHIRAI Y, et al. Organometal halide perovskites as visible-light sensitizers for photovoltaic cells. J. Am. Chem. Soc., 2009,131:6050-6051. |
| [2] | https://www.nrel.gov/pv/assets/pdfs/best-research-cell-efficiencies.20190923.pdf (2019). |
| [3] |
DONG Q, FANG Y J, SHAO Y C, et al. Electron-hole diffusion lengths >175 μm in solution-grown CH3NH3PbI3 single crystals. Science, 2015,347(6225):967-970.
URL PMID |
| [4] | LANG F, SHARGAIEVA O, BRUS V V, et al. Influence of radiation on the properties and the stability of hybrid perovskites. Adv. Mater., 2018,30(3):172905. |
| [5] | YANG Z, ZHONG M, LIANG Y, et al. SnO2-C60 pyrrolidine tris-acid (CPTA) as the electrontransport layer for highly efficient and stable planar Sn-based perovskite solar cells. Adv. Funct. Mater., 2019,29(42):1903621. |
| [6] | JIANG Q, ZHANG L, WANG H, et al. Enhanced electron extraction using SnO2 for high-efficiency planar-structure HC(NH2)2PbI3-based perovskite solar cells. Nat. Energy, 2017,2:16177. |
| [7] | XIONG L B, QIN M C, YANG G, et al. Performance enhancement of high temperature SnO2-based planar perovskite solar cells: electrical characterization and understanding of the mechanism. J. Mater. Chem., 2016,A4(21):8374-8383. |
| [8] | JIANG Q, CHU Z M, WANG P Y, et al. Planar-structure perovskite solar cells with efficiency beyond 21%. Adv. Mater., 2017,29(46):1703852. |
| [9] | KIM H, SANG H I, PARK N G. Organolead halide perovskite: new horizons in solar cell research. J. Phys. Chem. C, 2014,118(11):5615-5625. |
| [10] |
YANG D, YANG R X, WANG K, et al. High efficiency planar-type perovskite solar cells with negligible hysteresis using EDTA-complexed SnO2. Nat. Commun., 2018,9:3239.
DOI URL PMID |
| [11] | ZHANG P, WU J, ZHANG T, et al. Perovskite solar cells with ZnO electron-transporting materials. Adv. Mater., 2018,30(3):1703737. |
| [12] |
GRÄTZEL M. The light and shade of perovskite solar cells. Nat. Mater., 2014,13(9):838-842.
DOI URL PMID |
| [13] | CHUEH C C, LI C Z, JEN A K Y. Recent progress and perspective in solution-processed interfacial materials for efficient and stable polymer and organometal perovskite solar cells. Energy Environ. Sci., 2015,8(4):1160-1189. |
| [14] | CHEN Y C, MENG Q, ZHANG L R, et al. SnO2-based electron transporting layer materials for perovskite solar cells: a review of recent progress. Journal of Energy Chemistry, 2019,35:144-167. |
| [15] | HU T, BECKER T, POURDAVOUD N, et al. Indium-free perovskite solar cells enabled by impermeable tin-oxide electron extraction layers. Adv. Mater., 2017,29(27):1606656. |
| [16] | CHEN H, LIU D, WANG Y, et al. Enhanced performance of planar perovskite solar cells using low-temperature solution-processed Al-doped SnO2 as electron transport layers. Nanoscale Res. Lett., 2017,12(238):1-6. |
| [17] | KE W J, FANG G J, LIU Q, et al. Low-temperature solution- processed tin oxide as an alternative electron transporting layer for efficient perovskite solar cells. J. Am. Chem. Soc., 2015,137(21):6730-6733. |
| [18] | WANG C L, XIAO C X, YU, Y,et al. Understanding and eliminating hysteresis for highly efficient planar perovskite solar cells. Adv. Energy Mater., 2017,7(17):1700414. |
| [19] | JIANG Q, ZHAO Y, ZHANG X, et al. Surface passivation of perovskite film for efficient solar cells. Nat. Photonics, 2019,13:460-466. |
| [20] | SAIDAMINOV M I, KIM J, JAIN A, et al. Suppression of atomic vacancies via incorporation of isovalent small ions to increase the stability of halide perovskite solar cells in ambient air. Nat. Energy, 2018,3(8):648-654. |
| [21] | 柯维俊. 基于高效电子传输层的钙钛矿太阳能电池研究. 武汉: 武汉大学博士学位论文, 2016. |
| [22] | LIU Q, ZHANG X, LI C Y, et al. Effect of tantalum doping on SnO2 electron transport layer via low temperature process for perovskite solar cells. Appl. Phys. Lett., 2019,115:143903. |
| [23] | RAHUL R J, ASIT P, ARJUN S, et al. Effect of tantalum doping in a TiO2 compact layer on the performance of planar spiro-OMeTAD free perovskite solar cells. J. Mater. Chem. A, 2018,6:1037-1047. |
| [24] |
TEBBY Z, UDDIN T, NICOLAS Y, et al. Low-temperature UV processing of nanoporous SnO2 layers for dye-sensitized solar cells. ACS Appl. Mater. Interfaces, 2011,3(5):1485-1491.
DOI URL PMID |
| [25] |
TRAN V H, AMBADE R B, AMBADE S B, et al. Low-temperature solution-processed SnO2 nanoparticles as cathode buffer layer for inverted organic solar cells. ACS Appl. Mater. Interfaces, 2017,9(2):1645-1653.
DOI URL PMID |
| [26] |
REN X, YANG D, YANG Z, et al. Solution-processed Nb:SnO2 electron transport layer for efficient planar perovskite solar cells. ACS Appl. Mater. Interfaces, 2017,9(3):2421-2429.
DOI URL PMID |
| [27] | JEON N J, NA H, JUNG E H, et al. A fluorene-terminated hole-transporting material for highly efficient and stable perovskite solar cells. Nat. Energy, 2018,3:682-689. |
| [28] | LEE Y, PAEK S, CHO K T, et al. Enhanced charge collection with passivation of the tin oxide layer in planar perovskite solar cells. J. Mater. Chem. A, 2017,5:12729-12734. |
| [29] | LEE Y, LEE S, SEO G, et al. Efficient planar perovskite solar cells using passivated tin oxide as an electron transport layer. Adv. Sci., 2018,5(6):1800130. |
| [30] |
XING G, MATHEWS N, SUN S, et al. Long-range balanced electron- and hole-transport lengths in organic-inorganic CH3NH3PbI3. Science, 2013,342(6156):344-347.
DOI URL PMID |
| [31] | YANG D, YANG R, ZHANG J, et al. High efficiency flexible perovskite solar cells using superior low temperature TiO2. Energy Environ. Sci., 2015,8:3208-3214. |
| [32] | YANG S, YUE W, ZHU J, et al. Graphene-based mesoporous SnO2 with enhanced electrochemical performance for lithium-ion batteries. Adv. Funct. Mater., 2013,23(28):3570-3576. |
| [33] | MAHMUD M A, ELUMALAI N K, UPAMA M B, et al. Single vs mixed organic cation for low temperature processed perovskite solar cells. Electrochim. Acta., 2016,222(20):1510-1521. |
| [34] | WANG W, ZHANG Z, CAI Y, et al. Enhanced performance of CH3NH3PbI3-xClx perovskite solar cells by CH3NH3 modification of TiO2-perovskite layer interface. Nanoscale Res. Lett., 2016,11:316. |
| [35] | YU Z, CHEN B, LIU P, et al. Stable organic-inorganic perovskite solar cells without hole-conductor layer achieved via cell structure design and contact engineering. Adv. Funct. Mater., 2016,26(27):4866-4873. |
| [36] |
BAG M, RENNA L A, ADHIKARI R Y, et al. Kinetics of ion transport in perovskite active layers and its implications for active layer stability. J. Am. Chem. Soc., 2015,137(40):13130-13137.
URL PMID |
| [37] | AZPIROZ J M, MOSCONI E, BISQUERT J, et al. Defect migration in methylammonium lead iodide and its role in perovskite solar cell operation. Energy Environ. Sci., 2015,7:2118-2127. |
| [1] | 张万文, 罗建强, 刘淑娟, 马建国, 张小平, 杨松旺. 氧化锆间隔层的低温喷涂制备及其三层结构钙钛矿太阳能电池应用性能[J]. 无机材料学报, 2023, 38(2): 213-218. |
| [2] | 杨新月, 董庆顺, 赵伟冬, 史彦涛. 基于对氯苄胺的2D/3D钙钛矿太阳能电池[J]. 无机材料学报, 2022, 37(1): 72-78. |
| [3] | 于守武, 赵泽文, 赵晋津, 肖淑娟, 师岩, 高存法, 苏晓, 胡宇翔, 赵智胜, 王婕, 王连洲. 新型光伏储电原位集成电池研究进展[J]. 无机材料学报, 2020, 35(6): 623-632. |
| [4] | 熊浩, 张渤昕, 贾巍, 张青红, 谢华清. 高分子PVP添加剂对钙钛矿太阳电池稳定性的提升[J]. 无机材料学报, 2019, 34(1): 96-102. |
| [5] | 姜文龙, 周 伟, 应纪飞, 杨铁莹, 高延敏. ZnO/GO纳米材料基热稳定钙钛矿太阳能电池[J]. 无机材料学报, 2017, 32(1): 96-100. |
| [6] | 刘 畅, 苑 帅, 张海良, 曹丙强, 吴莉莉, 尹龙卫. 铜膜碘化法制备p型CuI薄膜及其用作空穴传输层的反型钙钛矿电池性能[J]. 无机材料学报, 2016, 31(4): 358-364. |
| [7] | 王素卿, 张学军, 田艳红, 张 丽, 李雅东. 锂离子电池负极材料PAN-ACF/SnO2的研究[J]. 无机材料学报, 2013, 28(8): 836-840. |
| [8] | 蒋大洞, 郑学军, 龚跃球, 朱 哲, 彭金峰. 退火温度对Bi3.15(Eu0.7Nd0.15)Ti3O12铁电薄膜力学性能的影响[J]. 无机材料学报, 2013, 28(2): 131-135. |
| [9] | 毛 瑞, 郭 洪, 田冬雪, 杨项军, 王世雄, 陈 景. 滤纸为模板制备中空SnO2纳米管锂离子电池负极材料[J]. 无机材料学报, 2013, 28(11): 1213-1216. |
| [10] | 崔旭梅, 左承阳, 蓝德均, 王 军. TiO2/SnO2纳米晶膜的制备及其电学性能研究[J]. 无机材料学报, 2013, 28(11): 1233-1236. |
| [11] | 徐 爽, 杨 颖, 邬洪源, 江 超, 井立强, 史克英. 一维Pt/SnO2纳米纤维的制备及NOx气敏性研究[J]. 无机材料学报, 2013, 28(06): 584-588. |
| [12] | 刘铂洋, 张 旺, 何昭文, 张 荻. 基于蝶翅鳞片三维结构的Au/SnO2纳米复合材料制备及其表面增强拉曼散射性能研究[J]. 无机材料学报, 2012, 27(9): 917-922. |
| [13] | 徐 浩, 汤成莉, 张 倩, 延 卫. 微波法合成锑掺杂SnO2电极[J]. 无机材料学报, 2012, 27(6): 667-672. |
| [14] | 李红霞, 季振国, 席俊华. 退火温度对TiO2基电阻开关器件性能的影响[J]. 无机材料学报, 2012, 27(10): 1063-1067. |
| [15] | 陆扣留, 季振国, 孔 哲, 李红霞, 张 峻. 掺锑的纳米SnO2/WPU复合隔热涂层的制备与表征[J]. 无机材料学报, 2012, 27(10): 1117-1120. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||